
Chemistry, 05.05.2020 19:24 elpancho755
Balance the equation by inserting coefficients as needed. equation: ZnS + HBr -> ZnBr_{2} + H_{2}S ZnS + HBr ⟶ ZnBr 2 + H 2 S

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 06:30
Identify the missing numbers below to show the result of multiplying the numbers (1.6 × 10-19)(5.0 × 106) = c × 10d
Answers: 1

Chemistry, 22.06.2019 07:00
How heavy is thanos? a) 3000 lbs b) all of it c) the price of tea in china d) heavy enough
Answers: 2


Chemistry, 22.06.2019 15:50
How many moles of potassium hydroxide are needed to completely react with 2.94 moles of aluminum sulfate
Answers: 1
You know the right answer?
Balance the equation by inserting coefficients as needed. equation: ZnS + HBr -> ZnBr_{2} + H_{2}...
Questions



Biology, 16.10.2019 12:30

Social Studies, 16.10.2019 12:30



History, 16.10.2019 12:30

Biology, 16.10.2019 12:30

English, 16.10.2019 12:30

Social Studies, 16.10.2019 12:30





Biology, 16.10.2019 12:30


History, 16.10.2019 12:30

English, 16.10.2019 12:30


History, 16.10.2019 12:30

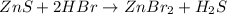
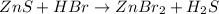
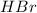 and we get the balanced chemical equation.
and we get the balanced chemical equation.

