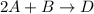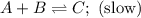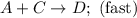Chemistry, 05.05.2020 20:16 jacksonyodell8601
Consider the mechanism. Step 1: A + B ⟶ C A+B⟶C slow Step 2: A + C ⟶ D A+C⟶D fast Overall: 2 A + B ⟶ D 2A+B⟶D Determine the rate law for the overall reaction, where the overall rate constant is represented as k .

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 06:00
Compare and contrast physical changes with chemical changes.
Answers: 3

Chemistry, 22.06.2019 14:30
Is a pencil falling to the floor anon contact force, a force, or a contact force
Answers: 1


Chemistry, 23.06.2019 06:00
Is the flow of energy during vaporizing more like the flow during melting or during freezing
Answers: 1
You know the right answer?
Consider the mechanism. Step 1: A + B ⟶ C A+B⟶C slow Step 2: A + C ⟶ D A+C⟶D fast Overall: 2 A + B ⟶...
Questions


Health, 28.05.2021 03:00

Advanced Placement (AP), 28.05.2021 03:00

Mathematics, 28.05.2021 03:00


History, 28.05.2021 03:00


Mathematics, 28.05.2021 03:00

Mathematics, 28.05.2021 03:00

English, 28.05.2021 03:00



History, 28.05.2021 03:00


Advanced Placement (AP), 28.05.2021 03:00





![\text{Rate}=k[A]^2[B]](/tpl/images/0642/1478/15407.png)