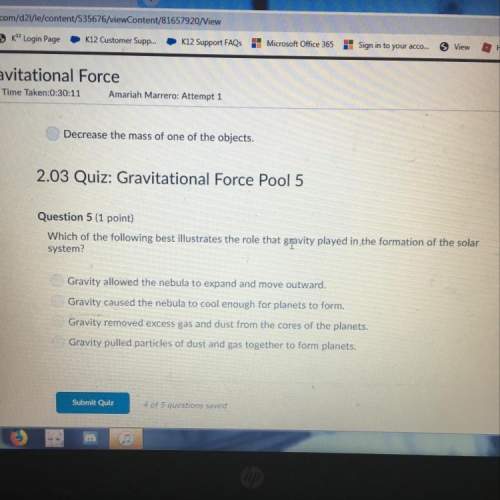
Which best explains why sodium is more reactive than magnesium?
A
Sodium has only one vale...

Chemistry, 05.05.2020 20:08 lucygperez7946
Which best explains why sodium is more reactive than magnesium?
A
Sodium has only one valence electron, while magnesium has two.
B
Sodium atoms typically have one more neutron than magnesium atoms do.
C
Sodium forms ions with a charge of 2+, but magnesium forms ions with a charge of 1+.
D
Sodium atoms tend to attract the electrons of other atoms more than magnesium atoms do.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 23:30
For the following dehydrohalogenation (e2) reaction, draw the zaitsev product(s) resulting from elimination involving c3–c4 (i.e., the carbon atoms depicted with stereobonds). show the product stereochemistry clearly. if there is more than one organic product, both products may be drawn in the same box. ignore elimination involving c3 or c4 and any carbon atom other than c4 or c3.
Answers: 3

Chemistry, 22.06.2019 02:50
Using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 pb+2(aq) + 2cl -(aq). if the value of ksp was determined to be only 1.2 x 10-2: too much solid has dissolved. additional precipitate is forming. the solution is unsaturated. the ions are now combining to reduce their concentrations.
Answers: 3

Chemistry, 22.06.2019 08:30
The mass of a neutron is equal to the mass of a proton plus the mass of an electron. true or false false true
Answers: 1

You know the right answer?
Questions

English, 15.07.2019 10:00

Mathematics, 15.07.2019 10:00



Mathematics, 15.07.2019 10:00

Biology, 15.07.2019 10:00








History, 15.07.2019 10:00

Mathematics, 15.07.2019 10:00



Biology, 15.07.2019 10:00