
Chemistry, 05.05.2020 22:20 raishagibson
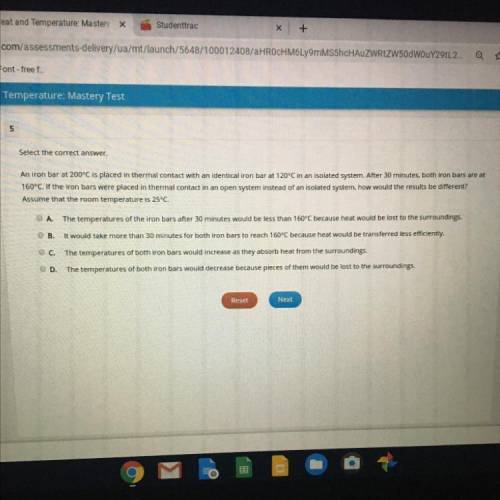
An iron bar at 200°C is placed in thermal contact with an identical iron bar at 120°C in an isolated system. After 30 minutes, both iron bars are at
160°C. If the iron bars were placed in thermal contact in an open system instead of an isolated system, how would the results be different?
Assume that the room temperature is 25°C.
A.
The temperatures of the iron bars after 30 minutes would be less than 160°C because heat would be lost to the surroundings.
B.
It would take more than 30 minutes for both iron bars to reach 160°C because heat would be transferred less efficiently.
C
D.
The temperatures of both iron bars would increase as they absorb heat from the surroundings.
The temperatures of both iron bars would decrease because pieces of them would be lost to the surroundings.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Omg imgonnafailnfiedkla use complete sentences to explain how the mass of hydrogen is conserved during cellular respiration.
Answers: 1


Chemistry, 22.06.2019 06:00
If you burn 10 kilograms of wood in a fire (combustion) what is the weight of the products after the fire has finished burning the wood?
Answers: 3

Chemistry, 22.06.2019 09:20
Give the orbital configuration of the phosphorus (p) atom.
Answers: 1
You know the right answer?
An iron bar at 200°C is placed in thermal contact with an identical iron bar at 120°C in an isolated...
Questions




Mathematics, 24.05.2021 18:20

Mathematics, 24.05.2021 18:20





Chemistry, 24.05.2021 18:20

History, 24.05.2021 18:20


Biology, 24.05.2021 18:20

History, 24.05.2021 18:20

Chemistry, 24.05.2021 18:20

English, 24.05.2021 18:20

Mathematics, 24.05.2021 18:20

Biology, 24.05.2021 18:20

History, 24.05.2021 18:20



