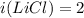Two aqueous solutions are prepared: 1.00 m Na2CO3 and 1.00 m LiCl. Which of the following statements is true?
A. The Na2CO3 solution has a higher osmotic pressure and higher vapor pressure than the LiCl solution.
B. The Na2CO3 solution has a higher osmotic pressure and higher boiling point than the LiCl solution.
C. The Na2CO3 solution has a lower osmotic pressure and lower vapor pressure than the LiCI solution.
D. The Na2CO3 solution has a lower osmotic pressure and higher boiling point than the LiCl solution.
E. None of these statements is true.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 14:50
Your roll: experienced electron speech is adressed to: a new "freshman class" of electrons job: write a speech task: you are to pretend that you are giving a speech to a new group of electrons. be sure to mention their placement in an atom, their charge, and their role in chemical bonding (ionic and covalent) be specific!
Answers: 3

Chemistry, 22.06.2019 11:30
Voltaic cells produce a positive overall charge. what does this indicate? a. the reaction is likely to be endothermic. b. the reaction is spontaneous. c. the reaction is not likely to occur. d. the reaction is not spontaneous.
Answers: 3

Chemistry, 22.06.2019 23:00
What is the energy in joules of a mole of photons associated with visible light of wavelength 486 nm?
Answers: 3

Chemistry, 23.06.2019 06:40
How many joules of heat are required to raise thetemperature of 750 g of water from 11.0 °c to 19.0 °c?
Answers: 1
You know the right answer?
Two aqueous solutions are prepared: 1.00 m Na2CO3 and 1.00 m LiCl. Which of the following statements...
Questions








Computers and Technology, 30.10.2019 00:31


Computers and Technology, 30.10.2019 00:31









Mathematics, 30.10.2019 00:31

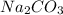 solution has a higher osmotic pressure and higher boiling point than LiCl solution.
solution has a higher osmotic pressure and higher boiling point than LiCl solution.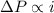 ,
, 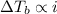 and
and 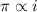
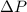 ,
, 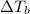 and
and 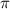 are lowering of vapor pressure, elevation in boiling point and osmotic pressure of solution respectively.
are lowering of vapor pressure, elevation in boiling point and osmotic pressure of solution respectively.  is van't hoff factor.
is van't hoff factor.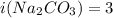 and
and