
Chemistry, 15.10.2019 18:40 dylankrenek
Butane, c4h10, reacts with oxygen, o2, to form water, h2o, and carbon dioxide, co2, as shown in the following chemical equation: 2c4h10(g)+13o2(g)-> 10h2o(g)+8co2(g)
calculate the mass of water produced when 1.77 grams of butane reacts with excessive oxygen?
calculate the mass of butane needed to produce 71.6 of carbon dioxide.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 20:30
The balanced chemical equation for this lab is: 3cucl2(aq) + 2al(s) 3cu(s) + 2alcl3(aq) if 10.5 g copper chloride react with 12.4 g aluminum, what is the limiting reactant?
Answers: 3

Chemistry, 22.06.2019 23:00
What is the mass of naoh that would have to be added to 500 ml of a solution of 0.20 m acetic acid in order to achieve a ph of 5.0?
Answers: 1

Chemistry, 23.06.2019 04:20
The equation below shows the reaction of zinc with hydrochloric acid (hcl). zn (s) + 2 hcl (aq) —> zncl2 (aq) + h2 (g) what will happen if the concentration of hcl is decreased? a. more zncl2 will be produced. b. the reaction rate will slow down. c. the hydrochloric acid will become more acidic. d. the reaction will produce water instead of hydrogen gas.
Answers: 1

Chemistry, 23.06.2019 09:00
What sources of error may have contributed to the percent yield not being 100 percent? think about things that may have led to inaccurate measurements or where mass of the product could have been lost if this experiment was conducted in a physical laboratory.
Answers: 2
You know the right answer?
Butane, c4h10, reacts with oxygen, o2, to form water, h2o, and carbon dioxide, co2, as shown in the...
Questions


English, 13.03.2021 14:00

Social Studies, 13.03.2021 14:00


English, 13.03.2021 14:00


Social Studies, 13.03.2021 14:00

Mathematics, 13.03.2021 14:00



Arts, 13.03.2021 14:00

Geography, 13.03.2021 14:00


Geography, 13.03.2021 14:00






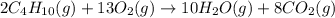
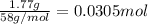
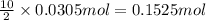 of water
of water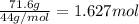
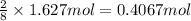 of butane
of butane

