Phosphoric acid is a triprotic acid with the following pKa values:
pKa1=2.148, pKa2=7.198, pKa3=12.375
You wish to prepare 1.000 L of a 0.0500 M phosphate buffer at pH 7.540. To do this, you choose to mix the two salt forms involved in the second ionization, NaH2PO4 and Na2HPO4, in a 1.000 L volumetric flask and add water to the mark. What mass of each salt will you add to the mixture?
a. Mass NaH2PO4
b. Mass Na2HPO4

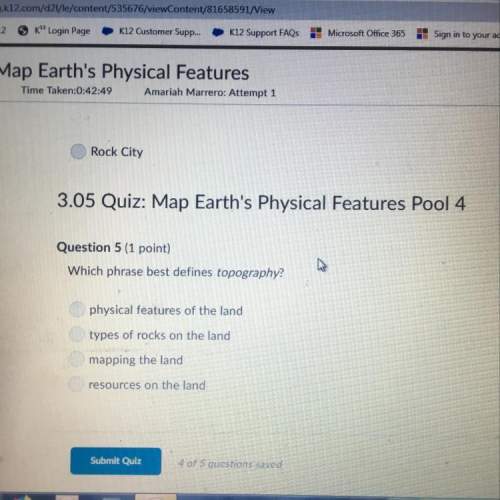
Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 21:40
It is important to wear proper protective equipment in lab even when not actively performing experiments because accidents can affect any researcher, even one not working on an experiment. select the best answer from the choices provided
Answers: 3

Chemistry, 21.06.2019 23:00
At room temperature what happens to the average kinetic energy of the molecules of a solid, liquid, and a gas
Answers: 2


Chemistry, 22.06.2019 11:00
Which statement is true about hcl? (5 points) select one: a. it is a salt because it increases the concentration of metallic ions. b. it is a salt because it is formed by the reaction of an acid and a base. c. it is an acid because it increases the concentration of hydroxyl ions. d. it is an acid because it increases the concentration of hydronium ions.
Answers: 1
You know the right answer?
Phosphoric acid is a triprotic acid with the following pKa values:
pKa1=2.148, pKa2=7.19...
pKa1=2.148, pKa2=7.19...
Questions










History, 15.12.2020 16:30




English, 15.12.2020 16:30



Biology, 15.12.2020 16:30