
When 40.0 mL of 1.00 M H2SO4 is added to 80.0 mL of 1.00 M NaOH at 20.00°C in a coffee cup calorimeter, the temperature of the aqueous solution increases to 29.20°C. If the mass of the solution is 120.0 g and the specific heat of the calorimeter and solution is 4.184 J/g • °C, how much heat is given off in the reaction?
Use q equals m C subscript p Delta T..
A). 4.62 kJ
B). 10.0 kJ
C). 14.7 kJ
D). 38.5 kJ

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 23:00
Esign techniques and materials that reduce the negative environmental impact of a structure are referred to as
Answers: 3

Chemistry, 22.06.2019 00:10
Select the correct answer. which phrase correctly describes temperature? o a. average rotational kinetic energy of the particles in an object o b. average energy of the particles in an object c. average translational kinetic energy of the particles in an object od. all energy possessed by the particles in an object
Answers: 1

Chemistry, 22.06.2019 03:30
In this chemical reaction, 325 grams of barium (ba) react completely. how many moles of lithium (li) are produced?
Answers: 1

Chemistry, 22.06.2019 21:00
Which answer tells the reason the earth’s climate is getting warmer? too many animals are becoming extinct. large glaciers are melting in antarctica. the earth is moving closer to the sun. driving cars gives off gases that trap heat in the atmosphere.
Answers: 1
You know the right answer?
When 40.0 mL of 1.00 M H2SO4 is added to 80.0 mL of 1.00 M NaOH at 20.00°C in a coffee cup calorimet...
Questions




Mathematics, 03.02.2021 03:50

Law, 03.02.2021 03:50



Social Studies, 03.02.2021 03:50


Mathematics, 03.02.2021 03:50

Social Studies, 03.02.2021 03:50

Mathematics, 03.02.2021 03:50

Mathematics, 03.02.2021 03:50


English, 03.02.2021 03:50




History, 03.02.2021 03:50

History, 03.02.2021 03:50

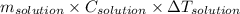 )
) is change in temperature.
is change in temperature.![[120.0g\times 4.184\frac{J}{g.^{0}\textrm{C}}\times (29.20-20.00)^{0}\textrm{C}]](/tpl/images/0648/3279/fdc1d.png)


