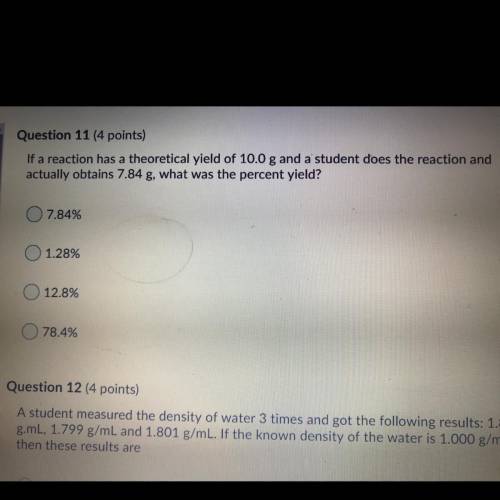
If a reaction has a theoretical yield of 10.0 g and a student does the reaction and
actually o...

Chemistry, 27.04.2020 01:39 marshallmattah
If a reaction has a theoretical yield of 10.0 g and a student does the reaction and
actually obtains 7.84 g, what was the percent yield?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:00
If the particles in a sample of matter have an orderly arrangement and move only in place, the sample is a
Answers: 1

Chemistry, 22.06.2019 02:10
3.) for each of the following compounds, draw the major organic product of reaction with hcl or naoh and circle whether the starting materials and products will be more soluble in organic solvent or water benzoic acid + hcl: benzoic acid + naoh: oh benzoic acid water/organic water organic fluorenone hс: fluorenone + naoh: fluorenone water/organic water/organic веnzocaine + hci: benzocaine + n»oh: h2n benzocaine water/organic water organic o=
Answers: 3


Chemistry, 22.06.2019 22:30
Which statement best summarizes the importance of ernest rutherford’s gold foil experiment? it proved that all of john dalton’s postulates were true. it verified j. j. thomson’s work on the atomic structure. it showed that an electron circles a nucleus in a fixed-energy orbit. it showed that a nucleus occupies a small part of the whole atom.
Answers: 1
You know the right answer?
Questions


History, 26.09.2019 03:00

Mathematics, 26.09.2019 03:00

Geography, 26.09.2019 03:00


Mathematics, 26.09.2019 03:00

Physics, 26.09.2019 03:00





English, 26.09.2019 03:00

Mathematics, 26.09.2019 03:00

Mathematics, 26.09.2019 03:00

Geography, 26.09.2019 03:00


Social Studies, 26.09.2019 03:00





