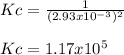Titanium and chlorine react to form titanium(IV) chloride, like this:
TiCl3 → Ti(s)+ 2Cl2(g)<...

Chemistry, 25.04.2020 04:26 rachelbrooks764
Titanium and chlorine react to form titanium(IV) chloride, like this:
TiCl3 → Ti(s)+ 2Cl2(g)
At a certain temperature, a chemist finds that a 5.2L reaction vessel containing a mixture of titanium, chlorine, and titanium(IV) chloride at equilibrium has the following composition:
Compound Amount
TiCl4 4.18g
Ti 1.32g
Cl2 1.08g
Calculate the value of the equilibrium constant Kc for this reaction.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 20:40
Astudent made the lewis dot diagram of a compound shown. what is the error in the lewis dot diagram? a)an o atom should transfer all of its six electrons to mg because the formula is mgo b) both electrons of mg should be transferred to one o adam because the formula is mgo c) the electrons should be transferred from each o add him to capital mg because mg has fewer electrons d) the number of dots around mg should be four because it has to transfer two electrons to each o
Answers: 1

Chemistry, 22.06.2019 09:00
At 300 mm hg, a gas has a volume of 380 l, what is the volume at standard pressure
Answers: 1

Chemistry, 22.06.2019 09:20
Give the orbital configuration of the phosphorus (p) atom.
Answers: 1

Chemistry, 22.06.2019 10:00
Nonpoint source pollution is difficult to control because it
Answers: 2
You know the right answer?
Questions

Mathematics, 12.10.2019 16:30

Biology, 12.10.2019 16:30


Social Studies, 12.10.2019 16:30

Social Studies, 12.10.2019 16:30

History, 12.10.2019 16:30


Mathematics, 12.10.2019 16:30



Mathematics, 12.10.2019 16:30

English, 12.10.2019 16:30

Physics, 12.10.2019 16:30



Mathematics, 12.10.2019 16:30


Mathematics, 12.10.2019 16:30

History, 12.10.2019 16:30

Mathematics, 12.10.2019 16:30

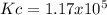
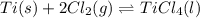
![Kc=\frac{1}{[Cl_2]^2}](/tpl/images/0626/7109/175a7.png)
![[Cl_2]_{eq}=\frac{1.08gCl_2*\frac{1molCl_2}{70.9gCl_2}}{5.2L}=2.93x10^{-3}M](/tpl/images/0626/7109/9b00a.png)