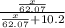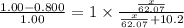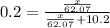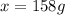Chemistry, 25.04.2020 04:06 amortegaa805
What mass of ethylene glycol, when mixed with 183 g H2O, will reduce the equilibrium vapor pressure of H2O from 1.00 atm to 0.800 atm at 100 °C? The molar masses of water and ethylene glycol are 18.02 g/mol and 62.07 g/mol, respectively. Assume ideal behavior for the solution.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 03:30
The boiling point of liquids is very high what does it indicate
Answers: 1

Chemistry, 22.06.2019 05:30
Liv sheldon given the balanced equation for an organic reaction: c2h2 + 2cl2 → c2h2cl4 this reaction is best classified as *
Answers: 1


Chemistry, 22.06.2019 13:00
Lab reagent, hypothesis test.a reference solution used as a lab reagent is purported to have a concentration of 5 mg/dl. six samples are taken from this solution and the following concentrations are recorded: (5.32, 4.88, 5.10, 4.73, 5.15, 4.75) mg/dl.these six measurements are assumed to be an srs of all possible measurements from solution.they are also assumed to have a standard deviation of 0.2, a normal distributin, and a mean concentration equal to the true concentration of the solution.carry out a significance test to determine whether these six measurements provide reliable evidence that the true concentration of the solution is actually not 5 mg/dl.
Answers: 1
You know the right answer?
What mass of ethylene glycol, when mixed with 183 g H2O, will reduce the equilibrium vapor pressure...
Questions



Biology, 17.11.2020 18:50

Health, 17.11.2020 18:50


History, 17.11.2020 18:50


English, 17.11.2020 18:50




English, 17.11.2020 18:50

Social Studies, 17.11.2020 18:50




Health, 17.11.2020 18:50


English, 17.11.2020 18:50

English, 17.11.2020 18:50

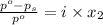
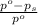 = relative lowering in vapor pressure
= relative lowering in vapor pressure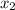 = mole fraction of solute =
= mole fraction of solute =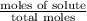
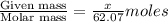
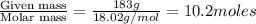
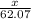 + 10.2
+ 10.2