Chemistry, 25.04.2020 03:50 abadir2008
Use the specific heat capacity that you calculated for granite to determine how many grams of granite at the initial temperature of 80 °C must mix with 3,000 g of water at the initial temperature of 20 °C to result in a final system temperature of 20.45 °X. (Hunt: Start by calculating how much heat energy is needed to change the water’s temperature by 0.45 °C). Show your work. Use the Gizmo to check your answer.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 09:00
What term is missing from the central region that describes hypotheses, theories, and laws? popular predictable mathematical falsifiable
Answers: 2

Chemistry, 22.06.2019 10:10
When water dissociates, each water molecule splits into a hydroxide ion and a) h 3 o + b) a hydrogen atom c) a hydrogen ion d) h 2 o e) oh —
Answers: 2

Chemistry, 22.06.2019 12:00
Ageochemist examines a piece of metal that he found in the soil. he performs tests to identify the metal from its density, electrical conductivity, and melting point. which statement best describes his investigation? a. he is determining physical properties that are sufficient to identify the metal.b. he is determining chemical properties that are sufficient to identify the metal.c. he is determining physical properties that are insufficient to identify the metal.d. he is determining chemical properties that are insufficient to identify the metal.
Answers: 3

Chemistry, 22.06.2019 17:10
Acalorimeter is to be calibrated: 51.203 g of water at 55.2 degree c is added to a calorimeter containing 49.783 g of water at 23.5c. after stirring and waiting for the system to equilibrate, the final temperature reached is 37.6 degree c. specific heat capacity of water (s = 4.18 j/g∙degree c). calculate the calorimeter constant. (smδt)warm water = -[(smδt)cold water + (calorimeterδtcold water)]
Answers: 2
You know the right answer?
Use the specific heat capacity that you calculated for granite to determine how many grams of granit...
Questions


Geography, 17.12.2020 21:20

Medicine, 17.12.2020 21:20


Mathematics, 17.12.2020 21:20

Mathematics, 17.12.2020 21:20

Mathematics, 17.12.2020 21:20

Chemistry, 17.12.2020 21:20



Mathematics, 17.12.2020 21:20



Mathematics, 17.12.2020 21:20

Mathematics, 17.12.2020 21:20




Mathematics, 17.12.2020 21:20

History, 17.12.2020 21:20

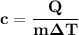
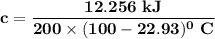
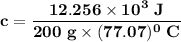
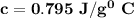
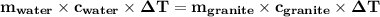
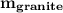 the subject of the formula;
the subject of the formula;