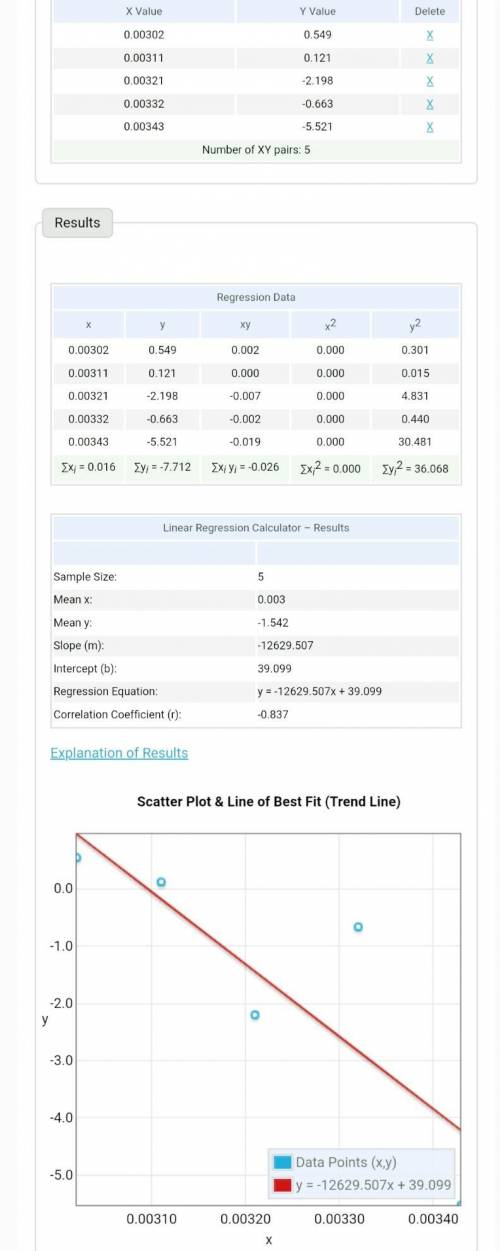
Plot your values of ln(Ksp) vs. 1/T and find the slope and y-intercept of the best fit line. Use the equation for the best fit line and the following equation l n ( K ) = − Δ H ∘ R T + Δ S ∘ R to calculate ΔH° and ΔS° for dissolving Borax (0/1pts) What is the slope of your best fit line in the plot? -7E-5 highlight_off (-1 pts) Incorrect. (0/1pts) What is ΔH° (kJ/mol)? .000582

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 03:30
In this chemical reaction, 325 grams of barium (ba) react completely. how many moles of lithium (li) are produced?
Answers: 1


Chemistry, 22.06.2019 21:00
Acandle’s wick is the fabric string that holds the flame, and it burns down at a constant slow pace when the candle is lit. the wick is usually surrounded by wax. which is the most important property of covalent compounds that makes them useful for making candle wax? a low boiling point a low melting point a high boiling point a high melting point
Answers: 1

Chemistry, 22.06.2019 21:00
In the experiment you asked to react hydrochloric acid and with sodium hydroxide. when measuring the volume of the reactants, which instrument would give the greatest precision.
Answers: 3
You know the right answer?
Plot your values of ln(Ksp) vs. 1/T and find the slope and y-intercept of the best fit line. Use the...
Questions

Mathematics, 26.01.2022 02:10

Chemistry, 26.01.2022 02:10





SAT, 26.01.2022 02:10

SAT, 26.01.2022 02:10

Computers and Technology, 26.01.2022 02:10



English, 26.01.2022 02:20


World Languages, 26.01.2022 02:20

Mathematics, 26.01.2022 02:20


Chemistry, 26.01.2022 02:20

Mathematics, 26.01.2022 02:20

Mathematics, 26.01.2022 02:20