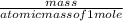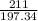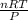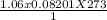1. Zinc metal reacts with 100 mL of 6.00 M H2SO4. How many grams of Zinc Sulfate are produced and how many liters of Hydrogen gas would be released?
2.A 211 g sample of BaCO3 is reacted with HNO3. Assuming the acid is present is excess, what mass and volume of CO2 gas will be produced?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 14:30
Janel’s class studied properties of matter and how matter can change. janel decided she would do an experiment mixing baking soda and vinegar.question: describe the properties of baking soda and vinegar, and explain the changes that janel should see when she mixes the two types of matter. •first, identify the physical state of matter of baking soda. describe another property of baking soda. •next, identify the physical state of matter of vinegar. describe another property of vinegar. •then, explain what janel should see when she mixes the baking soda and vinegar. •describe the states of matter of the new materials that are formed. •explain how janel can be certain a change has occurred. me
Answers: 3

Chemistry, 22.06.2019 10:30
Great amounts of electromagnetic energy from our sun and other bodies in space travel through space. which is a logical conclusion about these electromagnetic waves? their energy must be very their frequency must be very low these waves can travel without a medium they only travel through a vacuum of space
Answers: 2

Chemistry, 22.06.2019 13:30
In a ni-cd battery, a fully charged cell is composed of nickelic hydroxide. nickel is an element that has multiple oxidation states. assume the following proportions of the states: nickel charge proportions found 0 0.17 +2 0.3 +3 0.33 +4 0.5 (a) determine the mean of the nickel charge. enter the answer to 2 decimal places.(b) determine the cumulative distribution function of nickel charge.
Answers: 2

You know the right answer?
1. Zinc metal reacts with 100 mL of 6.00 M H2SO4. How many grams of Zinc Sulfate are produced and ho...
Questions


English, 27.01.2021 07:00

Mathematics, 27.01.2021 07:00


Chemistry, 27.01.2021 07:00






Biology, 27.01.2021 07:00


Social Studies, 27.01.2021 07:00

Mathematics, 27.01.2021 07:00



Mathematics, 27.01.2021 07:00



Chemistry, 27.01.2021 07:00

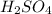 ⇒ ZnS
⇒ ZnS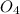 +
+ 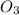 + 2HN
+ 2HN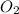 +
+ 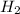 O
O