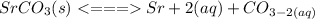Chemistry, 24.04.2020 18:19 brewerroserb
Consider the molar solubility of SrCO3 in 0.10 M Sr(NO3)2 versus in pure water. Ksp SrCO3 = 5.4 x 10-10 Which statement is true? A. The solubility of Sr(NO3)2 increases. B. The molar solubility of SrCO3 will be higher in 0.10 M 0 M Sr(NO3)2. C. The molar solubility of SrCO3 will be lower in water. D. The molar solubility of SrCO3 will be lower in 0.10 M 0 M Sr(NO3)2 because of Sr2+ E. There is not enough information given to determine an answer

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 10:00
The reactions shown here can be combined to make the overall reaction c(s) + h2o(g) ⇌ co(g) + h2(g) by reversing some and/or dividing all the coefficients by a number. a. c(s) + o2(g) → co2(g) k=1.363×10^69 b. 2 h2(g) + o2(g) → 2 h2o(g) k=1.389×10^80 c. 2co(g) + o2 (g) → 2 co2(g) k=1.477×10^90
Answers: 1

Chemistry, 22.06.2019 14:30
Which of the following represents the ester functional group? a. -coo- b. -cho c. -cooh d. c=o
Answers: 1

Chemistry, 22.06.2019 14:30
Select all that apply. using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 (s) pb+2(aq) + 2cl -(aq). the concentration of the products yield a ksp of 2.1 x 10-2:
Answers: 2

Chemistry, 22.06.2019 21:20
If a simple machine aduces the strength of a force, what must be increased? the speed of the input force the work the simple machine performs the size of the simple machine the distance over which the force is applied
Answers: 1
You know the right answer?
Consider the molar solubility of SrCO3 in 0.10 M Sr(NO3)2 versus in pure water. Ksp SrCO3 = 5.4 x 10...
Questions

Computers and Technology, 13.07.2019 20:00


Mathematics, 13.07.2019 20:00

Chemistry, 13.07.2019 20:00


Computers and Technology, 13.07.2019 20:00

Computers and Technology, 13.07.2019 20:00

Computers and Technology, 13.07.2019 20:00

Computers and Technology, 13.07.2019 20:00

Computers and Technology, 13.07.2019 20:00

Computers and Technology, 13.07.2019 20:00

Computers and Technology, 13.07.2019 20:00

Mathematics, 13.07.2019 20:00


Computers and Technology, 13.07.2019 20:00


Computers and Technology, 13.07.2019 20:00

Computers and Technology, 13.07.2019 20:00

Computers and Technology, 13.07.2019 20:00

Computers and Technology, 13.07.2019 20:00

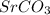 will be lower in 0.10 M 0 M
will be lower in 0.10 M 0 M 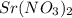 because of
because of 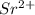 ) is the correct statement
) is the correct statement