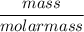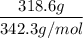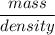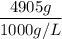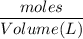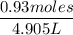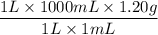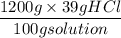A solution is prepared by dissolving 318.6 g sucrose (C12H22O11) in 4905 g of water. Determine the molarity of the solution
Commercial grade HCl solutions are typically 39.0% (by mass) HCl in water. Determine the molarity of the HCl, if the solution has a density of 1.20 g/mL.
Commercial grade HCl solutions are typically 39.0% (by mass) HCl in water. Determine the mol of the HCl, if the solution has a density of 1.20 g/mL.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 21:50
Liquid from a brewery fermentation contains 10% ethanol and 90% water. part of the fermentation product (50,000 kg/h) is pumped to a distillation column on the factory site. under current operating conditions, a distillate of 45% ethanol and 55% water is produced from the top of the column at a rate of one-tenth that of the feed. what is the composition of the waste "bottoms" from the still?
Answers: 2

Chemistry, 22.06.2019 23:10
Amines are good nucleophiles, even though they are neutral molecules. how would the rate of an sn2 reaction between an amine and an alkyl halide be affected if the polarity of the solvent is increased? amines are good nucleophiles, even though they are neutral molecules. how would the rate of an reaction between an amine and an alkyl halide be affected if the polarity of the solvent is increased? because both reactants in the rate-limiting step are neutral, the reaction will be faster if the polarity of the solvent is increased. because both reactants in the rate-limiting step are neutral, the reaction will be slower if the polarity of the solvent is increased. because both reactants in the rate-limiting step are neutral, the reaction will occur at the same rate if the polarity of the solvent is increased. request answer
Answers: 3


Chemistry, 23.06.2019 04:10
An unknown substance has been shown to have weak covalent bonds. which of the following is most likely a property of this substance? a. high ph b. high conductivity c. low melting point d. low flammability
Answers: 3
You know the right answer?
A solution is prepared by dissolving 318.6 g sucrose (C12H22O11) in 4905 g of water. Determine the m...
Questions


Mathematics, 02.10.2021 22:00


Mathematics, 02.10.2021 22:00


Mathematics, 02.10.2021 22:00

Arts, 02.10.2021 22:00

Business, 02.10.2021 22:00




Mathematics, 02.10.2021 22:00


Computers and Technology, 02.10.2021 22:00

Mathematics, 02.10.2021 22:00


Biology, 02.10.2021 22:00