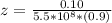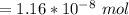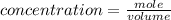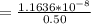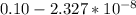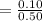Chemistry, 23.04.2020 01:17 gildedav001
A solution is prepared by adding 0.10 mole of Ni(NH3)6Cl2 to 0.50 L of 3.0 M NH3. Calculate [Ni(NH3)62 ] and [Ni2 ] in this solution. Koverall for Ni(NH3)62 is 5.5 x 108. That is, 5.5 x 108

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 03:30
At a temperature of 393 k, the temperature of a sample of nitrogen is 1.07 atm what will the pressure be at a temperature of 478 k
Answers: 1

Chemistry, 22.06.2019 04:00
Drag each label to the correct location on the chart. classify each reaction as endothermic or exothermic.
Answers: 1

Chemistry, 22.06.2019 05:00
When you mate two plants together the terms is called? answer it fast as possible plz! i have a test tomorrow!
Answers: 1
You know the right answer?
A solution is prepared by adding 0.10 mole of Ni(NH3)6Cl2 to 0.50 L of 3.0 M NH3. Calculate [Ni(NH3)...
Questions

Mathematics, 15.10.2019 07:10


Social Studies, 15.10.2019 07:10

Computers and Technology, 15.10.2019 07:10

Mathematics, 15.10.2019 07:10


Mathematics, 15.10.2019 07:10


Social Studies, 15.10.2019 07:10


Mathematics, 15.10.2019 07:10



Mathematics, 15.10.2019 07:10

Chemistry, 15.10.2019 07:10

History, 15.10.2019 07:10

Chemistry, 15.10.2019 07:10

Biology, 15.10.2019 07:10

Physics, 15.10.2019 07:10

![[Ni^{2+}]= 2.327*10^{-8} M](/tpl/images/0620/1201/754f2.png)
![[Ni(NH_3)_6]^{2+} _{(aq)}]= 0.3 M](/tpl/images/0620/1201/266ad.png)
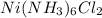 is
is 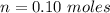
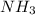 is =
is = 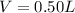
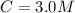
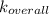 for
for ![[Ni(NH_3) _6^{2+}]](/tpl/images/0620/1201/584c3.png) is
is 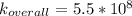
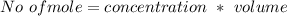
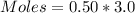
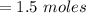
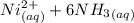 ⇄
⇄ ![[Ni(NH_3)_6]^{2+} _{(aq)}](/tpl/images/0620/1201/fd544.png)
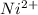
![0.10moles [Ni ^{2+}] * \frac{6moles NH_3}{1 mole [N_i^{2+}]}](/tpl/images/0620/1201/88989.png)
![= 0.6 moles [Ni^{2+}]](/tpl/images/0620/1201/c903d.png)
![1.5 moles [NH_3] * \frac{6moles NH_3}{1 mole [N_i^{2+}]}](/tpl/images/0620/1201/a0abd.png)
![= 9 moles [NH_3]](/tpl/images/0620/1201/cec0c.png)
![0.10 [Ni^{2+}] *\frac{1 moles [Ni(NH_3)_6]^{2+}}{1 mol [Ni^{2+}]}](/tpl/images/0620/1201/cfb14.png)
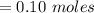
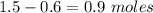
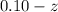
![K_{overall} = \frac{[Ni (NH_3) _6]^{2+}}{[Ni^{2+} [NH_3] ^6]}](/tpl/images/0620/1201/c7d6a.png)
![5.5*10^{8} = \frac{0.10 - z}{[z] [0.9 + 6z]^6}](/tpl/images/0620/1201/31e23.png)
![5.5*10^{8} = \frac{0.10}{[z] [0.5]^6}](/tpl/images/0620/1201/7e13e.png)