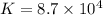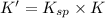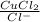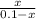The copper(I) ion forms a chloride salt (CuCl) that has Ksp = 1.2 x 10-6. Copper(I) also forms a complex ion with Cl-:Cu+ (aq) + 2Cl- (aq) ⇄ CuCl2- (aq) K = 8.7 x 104(a) Calculate the solubility of CuCl in pure water. (Ignore CuCl2- formation for part a).(b) Calculate the solubility of CuCl in 0.100 M NaCl solution.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 21:30
An alcohol thermometer makes use of alcohol's changing in order to measure temperature. as the temperature goes up, the alcohol contained in the thermometer increases in volume, filling more of the thermometer's tube.
Answers: 3

Chemistry, 22.06.2019 10:00
Ahydrogen atom has 1 electron. how many bonds can hydrogen form? a) 1 b) 2 c) 3 d) 4 e) 5
Answers: 3

Chemistry, 22.06.2019 21:00
One similarity and one difference between an element and a mixture of elements
Answers: 1

Chemistry, 22.06.2019 23:30
Aweight lifter raises a 1600 n barbell to a height of 2.0 meters. how much work was done? w = fd a) 30 joules b) 3000 joules c) 320 joules d) 3200 joules
Answers: 2
You know the right answer?
The copper(I) ion forms a chloride salt (CuCl) that has Ksp = 1.2 x 10-6. Copper(I) also forms a com...
Questions

History, 04.10.2019 19:00



Social Studies, 04.10.2019 19:00

Biology, 04.10.2019 19:00

Business, 04.10.2019 19:00

Mathematics, 04.10.2019 19:00


Biology, 04.10.2019 19:00



History, 04.10.2019 19:00


History, 04.10.2019 19:00

History, 04.10.2019 19:00

Computers and Technology, 04.10.2019 19:00

Health, 04.10.2019 19:00

Mathematics, 04.10.2019 19:00

Mathematics, 04.10.2019 19:00

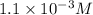 .
.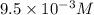 .
.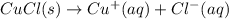
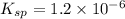
![K_{sp} = [Cu^{+}][Cl^{-}]](/tpl/images/0619/9671/6e8cf.png)
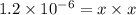
 ,
,