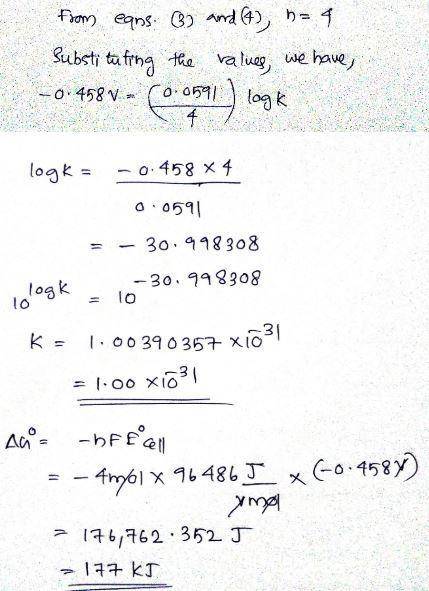Chemistry, 22.04.2020 21:00 Lydiaxqueen
Determine ∘ , Δ∘ , and K for the overall reaction from the balanced half-reactions and their standard reduction potentials. 2Co3+H3AsO3+H2O ↽−−⇀ 2Co2++H3AsO4+2H+Co3+e−↽−−⇀ Co2+ ∘=1.92H3AsO4+2H+2e−↽−−⇀ H3AsO3+H2O∘=0.575∘= ?K= ?Δ∘= ?Is the overall reaction spontaneous as written? not spontaneous spontaneousDetermine ∘E∘, Δ∘ΔG∘, and KK for the overall reaction from the balanced half-reactions and their standard reduction potentials.4Fe3++2H2O↽−−⇀ 4Fe2++O2+4H+Fe3++e−↽−−⇀ Fe2+ ∘=0.771 V1/2O2+2H++2e− ↽−⇀ H2O∘=1.229V∘=K=Δ∘=Is the overall reaction spontaneous as written?
A. spontaneous
B. not spontaneous

Answers: 3
Another question on Chemistry


Chemistry, 21.06.2019 22:50
Blank allows you to do calculations for situations in which only the amount of gas is constant a)boyle's law b)combined gas law c)ideal gas law d)dalton's law
Answers: 1

Chemistry, 21.06.2019 23:00
Plz choose one of the compounds from the table and explain how you know the numbers of atoms in your formula. is it possible for two different compounds to be made from the exact same two elements? why or why not? with a limited number of elements (less than 120 are known), does this mean we also have a small number of compounds or do we have a large number of compounds in this world?
Answers: 1

Chemistry, 22.06.2019 09:00
At 300 mm hg, a gas has a volume of 380 l, what is the volume at standard pressure
Answers: 1
You know the right answer?
Determine ∘ , Δ∘ , and K for the overall reaction from the balanced half-reactions and their standar...
Questions

History, 08.02.2021 07:10

English, 08.02.2021 07:10





History, 08.02.2021 07:10

Mathematics, 08.02.2021 07:10

English, 08.02.2021 07:10



Biology, 08.02.2021 07:10

English, 08.02.2021 07:10

Mathematics, 08.02.2021 07:20

History, 08.02.2021 07:20


Social Studies, 08.02.2021 07:20

Geography, 08.02.2021 07:20


Mathematics, 08.02.2021 07:20