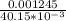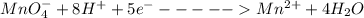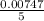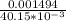Chemistry, 22.04.2020 04:39 aroland1990x
A Cu2+ solution is prepared by dissolving a 0.4749 g piece of copper wire in acid. The solution is then passed through a Walden reductor, reducing Cu2+ to Cu+ . The resulting Cu+ solution required 40.15 mL of each of the titrants to reach the endpoint. Calculate the concentration of each titrant.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 01:30
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. the values of phosphorous acid are 1.30 6.70 calculate the ph for each of the given points in the titration of 50.0 ml of 1.5 m h3po3(aq) with 1.5 m koh(aq) .
Answers: 3


Chemistry, 22.06.2019 05:30
Describe the interaction that occurs between two objects with the same electrical charge.
Answers: 1

You know the right answer?
A Cu2+ solution is prepared by dissolving a 0.4749 g piece of copper wire in acid. The solution is t...
Questions

English, 29.08.2020 07:01



Mathematics, 29.08.2020 07:01




English, 29.08.2020 07:01



Computers and Technology, 29.08.2020 07:01

Mathematics, 29.08.2020 07:01




Mathematics, 29.08.2020 07:01

Health, 29.08.2020 07:01

Mathematics, 29.08.2020 07:01


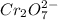 = 0.03101 M
= 0.03101 M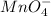 = 0.03721 M
= 0.03721 M
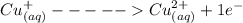
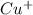 = 1 mole of
= 1 mole of 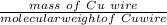
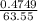
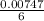
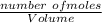
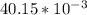 ; we have:
; we have: