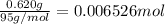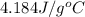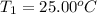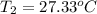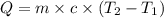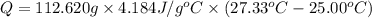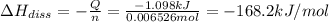Chemistry, 22.04.2020 03:29 milkshakegrande101
The salt magnesium chloride is soluble in water. When 0.620 g MgCl2 is dissolved in 112.00 g water, the temperature of the solution increases from 25.00 °C to 27.33 °C. Based on this observation, calculate the dissolution enthalpy, ΔdissH, of MgCl2. Assume that the specific heat capacity of the solution is 4.184 J g-1 °C-1 and that the energy transfer to the calorimeter is negligible. ΔdissH = kJ/mol

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 14:30
Heat flow inside earth activity i chose chemistry cause there is no science alert! the newspaper has been hacked, and all of the headlines have been changed to reflect old legends. it is your task to make sure they are corrected. choose one of the newspaper headlines. you will rewrite the headline and a one-to-two-paragraph article to make the news scientifically correct. be sure to include the following in your corrected newspaper article: new title reflecting correct information detailed information about the processes and tectonic plate interactions that are causing the geological event to occur real-world example of where to find the geological event on earth (picture and location) also review the grading rubric before you begin.
Answers: 1

Chemistry, 22.06.2019 15:30
The reactions of photosynthesis occur in the of plant cell? a.mitochondria. b. lysosomes. c. chloroplasts. d. chlorophyll
Answers: 1

Chemistry, 22.06.2019 17:30
What will most likely happen in the absence of a cell membrane? a) photosynthesis will not take place. b) the cell will not store food, water, nutrients, and waste. c) energy will not be released during cellular respiration. d) substances will pass in and out of the cell in an uncontrolled manner.
Answers: 1

You know the right answer?
The salt magnesium chloride is soluble in water. When 0.620 g MgCl2 is dissolved in 112.00 g water,...
Questions




Computers and Technology, 22.08.2019 19:10








Computers and Technology, 22.08.2019 19:10

Computers and Technology, 22.08.2019 19:10



Computers and Technology, 22.08.2019 19:10




Computers and Technology, 22.08.2019 19:10