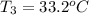Two humid streams are adiabatically mixed at 1 atm pressure to form a third stream. The first stream has a temperature of 40C, a relative humidity of 40%, and a volumetric flow rate of 3 L/s, while the second stream has a temperature of 15C, a relative humidity of 80%, and a volumetric flow rate of 1 L/s. Calculate the third stream’s temperature and relative humidity.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 05:00
What forms when chemical reactions combine pollution with sunlight?
Answers: 1

Chemistry, 22.06.2019 06:00
An atom of lithium (li) and an atom of chlorine (cl) engage in a chemical reaction. which correctly describes the structure of the resulting chemical compound? hint: consider the class of each element. the chemical compound will have a network structure. the chemical compound will have triple bonds. the chemical compound will have a ball-and-stick structure. the chemical compound will have double bonds.
Answers: 2

Chemistry, 22.06.2019 16:10
Given the following equation: 2a1 + 3mgcl2 --> 2alcl3 + 3mg how many moles of aluminum chloride are produced from 2.5 moles of magnesium chloride?
Answers: 1

Chemistry, 22.06.2019 18:10
The atom fluorine generally will become stable through the formation of an ionic chemical compound by accepting electron(s) from another atom. this process will fill its outer energy level of electrons.
Answers: 1
You know the right answer?
Two humid streams are adiabatically mixed at 1 atm pressure to form a third stream. The first stream...
Questions

Computers and Technology, 10.12.2021 21:00


History, 10.12.2021 21:00

Business, 10.12.2021 21:00

Geography, 10.12.2021 21:00

Social Studies, 10.12.2021 21:00

Mathematics, 10.12.2021 21:00



Mathematics, 10.12.2021 21:00

Computers and Technology, 10.12.2021 21:00

Mathematics, 10.12.2021 21:00

Social Studies, 10.12.2021 21:00

Mathematics, 10.12.2021 21:00

Business, 10.12.2021 21:00

Social Studies, 10.12.2021 21:00


Mathematics, 10.12.2021 21:10


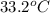 and its relative humidity is 50%.
and its relative humidity is 50%.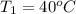 , R.H = 40%
, R.H = 40%
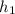 = 89 kj/kg
,
= 89 kj/kg
, 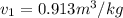
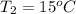
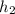 = 36.5 kj/kg
= 36.5 kj/kg
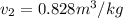
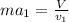
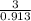
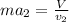
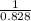
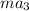 as follows.
as follows.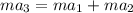
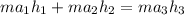
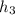 = 74.7855 kj/kg
= 74.7855 kj/kg
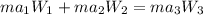
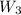 = 16.155
= 16.155
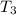 and
and 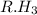 against
against