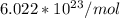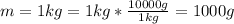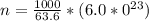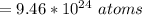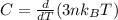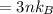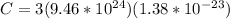Chemistry, 22.04.2020 02:51 journeywalker19
For most solids at room temperature, the specific heat is determined by oscillations of the atom cores in the lattice (each oscillating lattice site contributes 3kT of energy, by equipartition), as well as a contribution from the mobile electrons (if it's a metal). At room temperature the latter contribution is typically much smaller than the former, so we will ignore it here. In other words, you can reasonably estimate the specific heat simply by counting the number of atoms! Use this fact to estimate the specific heat of copper (atomic mass = 63.6), given that the specific heat of aluminum (atomic mass = 27.0) is 900 J/kg-K.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 23:00
Why are the trends and exceptions to the trends in ionization energy observed?
Answers: 1

Chemistry, 22.06.2019 12:00
What is the subscript for oxygen in its molecular formula
Answers: 1

Chemistry, 22.06.2019 17:00
The biosphere of the earth is made up of what compound? organic or inorganic?
Answers: 3

Chemistry, 22.06.2019 17:40
Which statement about hf is true? it is zero for any compound in its standard state. it is positive when the bonds of the product store more energy than those of the reactants. it is negative when a compound forms from elements in their standard states. it is zero for any element that is in the liquid state.
Answers: 1
You know the right answer?
For most solids at room temperature, the specific heat is determined by oscillations of the atom cor...
Questions


Mathematics, 19.03.2020 08:27


Biology, 19.03.2020 08:27






English, 19.03.2020 08:28



Mathematics, 19.03.2020 08:28




Mathematics, 19.03.2020 08:28


Mathematics, 19.03.2020 08:28

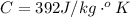
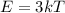
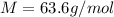
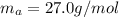
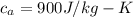
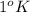
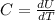
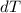 is the change in temperature
is the change in temperature 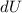 is the change in internal energy
is the change in internal energy 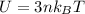
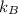 is the Boltzmann constant with a value of
is the Boltzmann constant with a value of 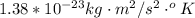
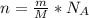
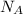 is the Avogadro's number with a constant value of
is the Avogadro's number with a constant value of