Chemistry, 21.04.2020 23:24 melidacampos12
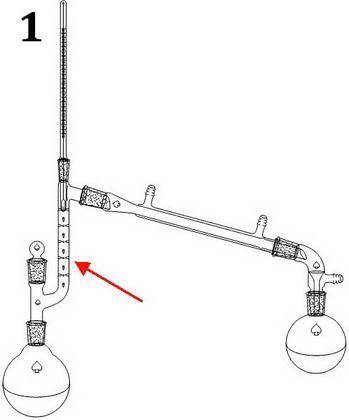
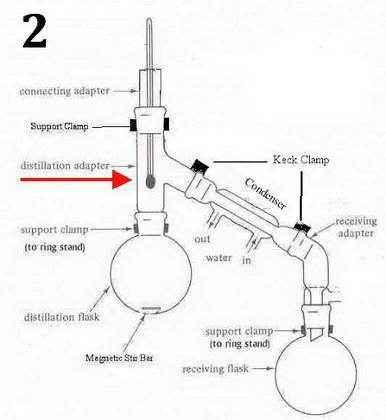
You carry out a reaction that yields a major product Q (boiling point: 120°C) and side product R (boiling point: 150°C). The ratio of Q to R is 70:30. Both Q and R are liquids. m) You use ethyl acetate (0.902 g/mL) to act as an extraction solvent. What are three main criteria for the liquid/liquid extraction organic solvent based on the lecture notes? (3 pts) What layer will ethyl acetate be at? (upper/lower) (1 pt) n) Q and R will be soluble in the organic layer (ethyl acetate) – any residual inorganic material was removed by water. Provide two ways you remove water from the organic layer. (2 pts) o) After the ethyl acetate is evaporated, Q and R (a mixture of liquids) remain. What criteria does this mixture have to satisfy to be separated by simple or fractional distillation (besides bp difference and composition)? (1 pt) p) What kind of distillation would you carry out (simple or fractional) and why? (3 pts) q) What parts of the microscale kit (seen on page 12 in the textbook) that you use for the distillation you pick that you don’t use for the alternate distillation (the one you didn’t pick)? (3 pts)

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 19:40
Scientists have developed an explanation of a phenomenon from several verified hypotheses. the explanation has been confirmed through numerous experimental tests.which option best describes this explanation? a. scientific lawb. research questionc. hypothesisd. scientific theory
Answers: 3

Chemistry, 22.06.2019 20:00
In vapor-liquid equilibrium in a binary mixture, both components are generally present in both phases. how many degrees of freedom are there for such a system? the reaction between nitrogen and hydrogen to form ammonia occurs in the gas phase. how many degrees of freedom are there for this system? steam and coal react at high temperatures to form hydrogen, carbon monoxide, carbon dioxide, and methane. the following reactions have been suggested as being involved in the chemical transformation:
Answers: 3

Chemistry, 22.06.2019 21:50
28. which is not a reason that water is used to store spent fuel rods from nuclear power plants? water increases the speed of the chain reaction in the fuel rods. water protects nuclear power plant workers from the high temperature and radiation of the fuel rods. water acts as a radiation shield to reduce the radiation levels. water cools the spent rods. salts action
Answers: 1

Chemistry, 22.06.2019 23:00
What is the average rate of the reaction between 10 and 20 s?
Answers: 1
You know the right answer?
You carry out a reaction that yields a major product Q (boiling point: 120°C) and side product R (bo...
Questions

Chemistry, 29.10.2019 08:31

Mathematics, 29.10.2019 08:31



Chemistry, 29.10.2019 08:31

English, 29.10.2019 08:31


Mathematics, 29.10.2019 08:31

Biology, 29.10.2019 08:31


Chemistry, 29.10.2019 08:31

Chemistry, 29.10.2019 08:31


English, 29.10.2019 08:31



Geography, 29.10.2019 08:31

Mathematics, 29.10.2019 08:31

History, 29.10.2019 08:31

Chemistry, 29.10.2019 08:31