Chemistry, 21.04.2020 23:01 blackops7959
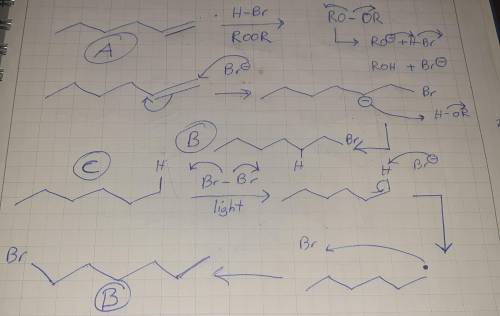
Stbank, Question 075 Get help answering Molecular Drawing questions. Compound A, C6H12 reacts with HBr/ROOR to give compound B, C6H13Br. Compound C, C6H14, reacts with bromine and light to produce compound B, C6H13Br. Suggest structures for compounds A, B, and C. Draw structure of the compound A. Edit Draw structure of the compound B. Edit Draw structure of the compound C.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 10:50
An atom of lithium-7 has an equal number of(1) electrons and neutrons(2) electrons and protons(3) positrons and neutrons(4) positrons and protons
Answers: 2



Chemistry, 22.06.2019 20:10
The lattice enthalpy (formation of ionic solid from ions in the gas phase) for agcl(s) is -916 kj/mol and the hydration enthalpy (dissolution of gaseous ions into water) is -850 kj/mol. how much heat (in joules) is involved in forming 1l of saturated agcl solution (1.8 × 10-4 g / 100 ml water) by dissolving agcl(s)? assume solution volume does not change much upon dissolution. the equations are given below. ag+(g) + cl−(g) æ agcl(s)
Answers: 3
You know the right answer?
Stbank, Question 075 Get help answering Molecular Drawing questions. Compound A, C6H12 reacts with H...
Questions

English, 17.06.2020 09:57



Mathematics, 17.06.2020 09:57


History, 17.06.2020 09:57



Mathematics, 17.06.2020 09:57


Social Studies, 17.06.2020 09:57



Mathematics, 17.06.2020 09:57

Spanish, 17.06.2020 09:57

Mathematics, 17.06.2020 09:57




Mathematics, 17.06.2020 09:57