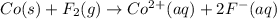Assign oxidation states to all of the species in the following redox reaction. For the reactants, identify electron loss or gain, the species oxidized, the species reduced, the oxidizing agent and the reducing agent. Co(s) + F2(g) Co2+(aq) + 2F-(aq) Oxidation state Electron loss or gain Oxidized or reduced Reducing or oxidizing agent

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 16:30
Asample of silver (with work function ? = 4.52 ev) is exposed to an ultraviolet light source (? = 200 nm), which results in the ejection of photoelectrons. what changes will be observed if: silver is replaced with copper (? = 5.10 ev) more photoelectrons ejected no photoelectrons are emitted fewer photoelectrons ejected more energetic photoelectrons (on average) less energetic photoelectrons (on average)
Answers: 3

Chemistry, 22.06.2019 05:30
The table describes how some substances were formed substance 19 description formed by boiling pure water formed by combining three hydrogen atoms to every nitrogen atom formed by adding 5 g of sugar to 1 l of water formed by compressing carbon under high pressure based on the given descriptions, which substance is most likely a mixture?
Answers: 1

Chemistry, 22.06.2019 05:30
Compare and contrast physical changes with chemical changes.
Answers: 1

Chemistry, 22.06.2019 07:00
At 450 mm hg a gas has a volume of 760 l, what is its volume at standard pressure
Answers: 2
You know the right answer?
Assign oxidation states to all of the species in the following redox reaction. For the reactants, id...
Questions

History, 24.02.2021 01:00

Mathematics, 24.02.2021 01:00

Mathematics, 24.02.2021 01:00

Biology, 24.02.2021 01:00

Mathematics, 24.02.2021 01:00


Mathematics, 24.02.2021 01:00



Geography, 24.02.2021 01:00

History, 24.02.2021 01:00

Mathematics, 24.02.2021 01:00

History, 24.02.2021 01:00

Mathematics, 24.02.2021 01:00

History, 24.02.2021 01:00



Mathematics, 24.02.2021 01:00


English, 24.02.2021 01:00

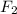 gain two electrons and thus gets reduced and acts as oxidizing agent.
gain two electrons and thus gets reduced and acts as oxidizing agent.