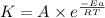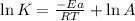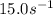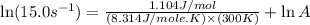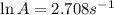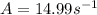A chemical reaction has an energy of activation Ea = 1∙104 J mol-1 at T = 300 K. The first-order rate constant for this reaction was found to be 15.0 s-1. In the presence of a catalyst, the activation energy is reduced to 1∙103 J mol-1. Calculate the pre-exponential factor in the Arrhenius equation

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:00
Write a hypothesis that answers the lesson question, “while observing a chemical reaction, how can you tell which reactant is limiting? ” hypothesis: if a substance is the limiting reactant, then . . because . .
Answers: 1

Chemistry, 22.06.2019 09:50
What are four significant sources of ghgs that come from wostem washington?
Answers: 2

Chemistry, 22.06.2019 16:00
Which of the following is the correct definition of chemical energy? a. energy an object has because of its motion or position b. energy resulting from the flow of charged particles, such as electrons or ions c. energy produced from the splitting of atoms d. energy stored in chemical bonds of molecules
Answers: 1

Chemistry, 22.06.2019 17:40
How much heat is added if 0.814g of water increase in temperature by 0.351 degree c?
Answers: 3
You know the right answer?
A chemical reaction has an energy of activation Ea = 1∙104 J mol-1 at T = 300 K. The first-order rat...
Questions






Mathematics, 08.03.2021 21:10



Geography, 08.03.2021 21:10




Mathematics, 08.03.2021 21:10


Mathematics, 08.03.2021 21:10

Advanced Placement (AP), 08.03.2021 21:10

Mathematics, 08.03.2021 21:10

Mathematics, 08.03.2021 21:10

Computers and Technology, 08.03.2021 21:10