
A chemistry student needs 75 g of thiophene foran experiment. He has available 0.50 kg of a 29.3% w/w solution of thiophene in carbon tetrachloride.
Calculate the mass of solution the student should use. Ifthere's not enough solution, press the "No solution" button.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 10:00
Why is the structure of molecule important to its function?
Answers: 1

Chemistry, 22.06.2019 11:40
Modern pennies are composed of zinc coated with copper. a student determines the mass of a penny to be 2.482 g and then makes several scratches in the copper coaling (to expose the underlying zinc). the student puts the scratched penny in hydrochloric acid, where the following reaction occurs between the zinc and the hcl (the copper remains undissolved): zn(s) + 2 hcl(aq) → h2(g) + zncl(aq)the student collects the hydrogen produced over water at 25 °c. the collected gas occupies a volume of 0.899 l at a total pressure of 79 j mmhg. calculate the percent zinc (by mass) in the penny. (assume that all the zn in the penny dissolves.)
Answers: 1

Chemistry, 22.06.2019 13:00
Adepositional also feature that forms where a stream enters a lake or an ocean is a
Answers: 2

Chemistry, 22.06.2019 15:00
20 pts ‼️ an unmanned spacecraft travels to mars. mars has a lower strength of gravity than earth. where in the image is the spacecraft’s weight the greatest?
Answers: 2
You know the right answer?
A chemistry student needs 75 g of thiophene foran experiment. He has available 0.50 kg of a 29.3% w/...
Questions

Physics, 27.09.2019 08:00


Mathematics, 27.09.2019 08:00


Biology, 27.09.2019 08:00

Mathematics, 27.09.2019 08:00

Mathematics, 27.09.2019 08:00

Mathematics, 27.09.2019 08:00

Business, 27.09.2019 08:00



History, 27.09.2019 08:00


English, 27.09.2019 08:00

Mathematics, 27.09.2019 08:00


Health, 27.09.2019 08:00


Mathematics, 27.09.2019 08:00

Biology, 27.09.2019 08:00

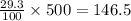 g of solute (1kg=1000g)
g of solute (1kg=1000g)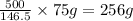 of solution
of solution

