Chemistry, 21.04.2020 18:20 Isabella1319
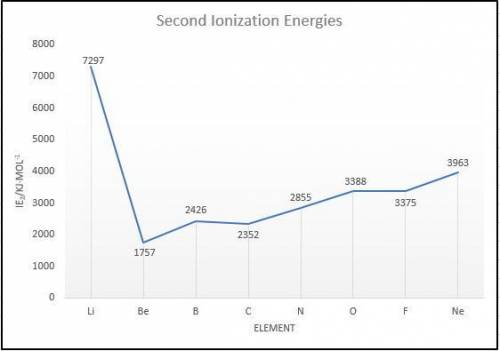
Elements in group 7A in the periodic table are called the halogens: elements in group 6A are called the chalcogens (a)
What is the most common oxidation state of the chalcogens compared to the halogens? (b) For each of the following
periodic properties. state whether the halogens or the chalcogens have larger values: atomic radii, ionic radii of the most
common oxidation state, first ionization energy. second ionization energy.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:30
Why is soap used to remove grease? a. its nonpolar end dissolves the grease. b. it makes the water bond with the grease. c. it chemically bonds with the grease. d. its polar end dissolves the grease.correct answer for apex - a, its nonpolar end dissolves the grease.
Answers: 1

Chemistry, 22.06.2019 14:30
How does a noncompetitive inhibitor reduce an enzyme’s activity?
Answers: 1

Chemistry, 22.06.2019 19:50
Which sentence from holes contains an implied personality trait? stanley and his parents had tried to pretend that he was just going away to camp for a while, just like rich kids do. he'd just been in the wrong place at the wrong time. stanley felt somewhat dazed as the guard unlocked his handcuffs and led him off the bus. stanley nodded to show he understood
Answers: 3

You know the right answer?
Elements in group 7A in the periodic table are called the halogens: elements in group 6A are called...
Questions

Mathematics, 22.02.2020 00:19

English, 22.02.2020 00:19


Mathematics, 22.02.2020 00:19


History, 22.02.2020 00:19









Social Studies, 22.02.2020 00:20

Mathematics, 22.02.2020 00:20



Chemistry, 22.02.2020 00:20