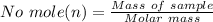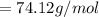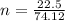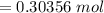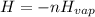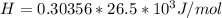Chemistry, 21.04.2020 18:07 roseemariehunter12
The following information is given for ether, C2H5OC2H5, at 1atm: boiling point = 34.6 °C Hvap(34.6 °C) = 26.5 kJ/mol specific heat liquid = 2.32 J/g°C /At a pressure of 1 atm, what is H in kJ for the process of condensing a 22.5 g sample of gaseous ether at its normal boiling point of 34.6 °C.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:00
14. many depressants reduce small muscle control, making it harder for a. you to steer b. your mind to consider complex problems c. the eye to scan, focus, or stay still d. the kidneys to filter alcohol out of the bloodstream
Answers: 3

Chemistry, 22.06.2019 05:30
Why is soap used to remove grease? a. its nonpolar end dissolves the grease. b. it makes the water bond with the grease. c. it chemically bonds with the grease. d. its polar end dissolves the grease.correct answer for apex - a, its nonpolar end dissolves the grease.
Answers: 1

Chemistry, 22.06.2019 18:00
How is energy related to the change of state represented by the model? atoms gain energy as a solid changes to a liquid. atoms gain energy as a solid changes to a gas. atoms lose energy as a solid changes to a liquid. atoms lose energy as a solid changes to a gas.
Answers: 3

Chemistry, 23.06.2019 03:00
What volume does 1.70 ×10–3 mol of chlorine gas occupy if its temperature is 20.2 °c and its pressure is 795 mm hg?
Answers: 3
You know the right answer?
The following information is given for ether, C2H5OC2H5, at 1atm: boiling point = 34.6 °C Hvap(34.6...
Questions



Mathematics, 06.06.2020 05:58



Biology, 06.06.2020 05:58








Physics, 06.06.2020 05:58



History, 06.06.2020 05:58

Mathematics, 06.06.2020 05:58


Chemistry, 06.06.2020 05:58

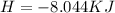
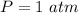
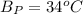
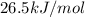

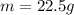
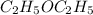 is given as
is given as