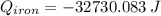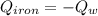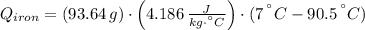Chemistry, 21.04.2020 17:37 kaylaanderson348
A piece of iron with a mass of 56.2 grams is heated and placed into a calorimeter containing 93.64 grams of water at 7.0 degrees Celsius. The final temperature of the water and the iron is 90.5 degrees Celsius. Assuming no heat is lost to the surroundings, how much heat (in Joules) does the iron release? Round your answer to the nearest 0.1 Joules.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 00:20
What are the spectator ions in 2h+ + so42- + ca2+ + 2r → caso4 + 2h+ + 21?
Answers: 1

Chemistry, 22.06.2019 11:40
Calculate the number of kilojoules to warm 125 g of iron from 23.5°c to 78.0°c.
Answers: 3

Chemistry, 22.06.2019 22:00
How many moles of no2 will form when 3.3 moles of cu are reacted with excess hno3?
Answers: 3

Chemistry, 23.06.2019 00:40
To prevent the presence of air, noble gases are placed over highly reactive chemicals to act as inert "blanketing" gases. a chemical engineer places a mixture of noble gases consisting of 4.37 g of he, 13.36 g of ne, and 36.65 g of kr in a piston-cylinder assembly at stp. calculate the partial pressure in torr of kr.
Answers: 1
You know the right answer?
A piece of iron with a mass of 56.2 grams is heated and placed into a calorimeter containing 93.64 g...
Questions

Spanish, 28.01.2020 17:40


Mathematics, 28.01.2020 17:40


Mathematics, 28.01.2020 17:40





Mathematics, 28.01.2020 17:40

Mathematics, 28.01.2020 17:40

English, 28.01.2020 17:40


History, 28.01.2020 17:40