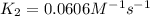Chemistry, 21.04.2020 17:00 noeltan12031
The reaction between nitrogen dioxide and carbon monoxide is NO2(g)+CO(g)→NO(g)+CO2(g)NO2(g)+CO( g)→NO(g)+CO2(g) The rate constant at 701 KK is measured as 2.57 M−1⋅s−1M−1⋅s−1 and that at 895 KK is measured as 567 M−1⋅s−1M−1⋅s−1. The activation energy is 1.5×102 1.5×102 kJ/molkJ/mol. Predict the rate constant at 525 KK .

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 19:00
Asyringe contains 56.05 ml of gas at 315.1 k. what volume will that gas occupy if the temperature is increased to 380.5 k? a) 12.41 b) 46.42 c) 67.68 d) 81.74
Answers: 1

Chemistry, 22.06.2019 09:00
If you chip a tooth, most likely you will go to the dentist to have the missing material filled in. currently the material used to fill in teeth is a polymer that is flexible when put in, yet is hardened to the strength of a tooth after irradiation with blue light at a wavelength of 461 nm. what is the energy in joules for a photon of light at this wavelength?
Answers: 1

Chemistry, 22.06.2019 09:30
One way that radioactive waste is treated is by burying it in repositories. the repositories are found only in states with very low populations. true or false? a. trueb. false(also i meant to put high school but it put down middle school instead)
Answers: 1

You know the right answer?
The reaction between nitrogen dioxide and carbon monoxide is NO2(g)+CO(g)→NO(g)+CO2(g)NO2(g)+CO( g)→...
Questions



Mathematics, 06.10.2020 01:01

Mathematics, 06.10.2020 01:01




Mathematics, 06.10.2020 01:01

Mathematics, 06.10.2020 01:01


English, 06.10.2020 01:01





Computers and Technology, 06.10.2020 01:01

Mathematics, 06.10.2020 01:01


History, 06.10.2020 01:01

Mathematics, 06.10.2020 01:01

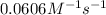
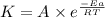
![\log (\frac{K_2}{K_1})=\frac{Ea}{2.303\times R}[\frac{1}{T_1}-\frac{1}{T_2}]](/tpl/images/0614/9250/6d953.png)
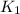 = rate constant at
= rate constant at 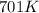 =
= 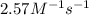
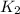 = rate constant at
= rate constant at 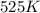 = ?
= ?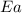 = activation energy for the reaction =
= activation energy for the reaction = 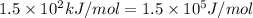
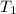 = initial temperature = 701 K
= initial temperature = 701 K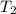 = final temperature = 525 K
= final temperature = 525 K![\log (\frac{K_2}{2.57M^{-1}s^{-1}})=\frac{1.5\times 10^5J/mol}{2.303\times 8.314J/mole.K}[\frac{1}{701K}-\frac{1}{525K}]](/tpl/images/0614/9250/4adff.png)