
Chemistry, 21.04.2020 16:16 Crtive5515
A student has a 2.66 L bottle that contains a mixture of O2 , N2 , and CO2 with a total pressure of 4.50 bar at 298 K . She knows that the mixture contains 0.297 mol N2 and that the partial pressure of CO2 is 0.269 bar . Calculate the partial pressure of O2 .

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:40
How many electrons does silver have to give up in order to achieve a sido noble gas electron configuration
Answers: 1

Chemistry, 22.06.2019 05:30
What type of reaction is shown below? check all that apply. 2h2o2 → 2h2o + o2 synthesis decomposition combustion
Answers: 1

Chemistry, 22.06.2019 21:20
One way in which the useful metal copper is produced is by dissolving the mineral azurite, which contains copper(ii) carbonate, in concentrated sulfuric acid. the sulfuric acid reacts with the copper(ii) carbonate to produce a blue solution of copper(ii) sulfate. scrap iron is then added to this solution, and pure copper metal precipitates out because of the following chemical reaction: (s) (aq) (s) (aq) suppose an industrial quality-control chemist analyzes a sample from a copper processing plant in the following way. he adds powdered iron to a copper(ii) sulfate sample from the plant until no more copper will precipitate. he then washes, dries, and weighs the precipitate, and finds that it has a mass of .
Answers: 2

You know the right answer?
A student has a 2.66 L bottle that contains a mixture of O2 , N2 , and CO2 with a total pressure of...
Questions

History, 19.11.2020 05:20

History, 19.11.2020 05:20

Biology, 19.11.2020 05:20



Geography, 19.11.2020 05:20

Mathematics, 19.11.2020 05:20

Mathematics, 19.11.2020 05:20

Mathematics, 19.11.2020 05:20

Mathematics, 19.11.2020 05:20




Biology, 19.11.2020 05:20

Mathematics, 19.11.2020 05:20

Mathematics, 19.11.2020 05:20

English, 19.11.2020 05:20

Mathematics, 19.11.2020 05:20

Spanish, 19.11.2020 05:20

Spanish, 19.11.2020 05:20

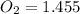 bar.
bar.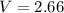 L
L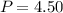 bar
bar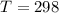 K
K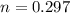 mole
mole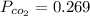 bar
bar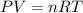
 = gas constant
= gas constant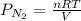
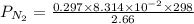
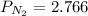 bar
bar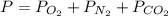
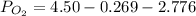
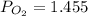 bar
bar

