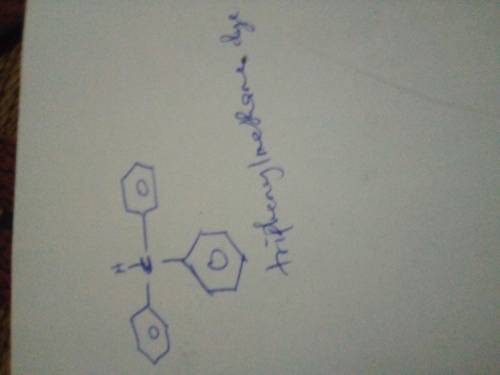
Triphenylmethanol is insoluble in water, but when it is treated with concentrated H2SO4, a bright yellow solution results. As this yellow solution is diluted with water, its color disappears, and a precipitate of triphenylmethanol reappears. Suggest a structure for the bright yellow species.

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 08:30
The mass of a neutron is equal to the mass of a proton plus the mass of an electron. true or false false true
Answers: 1

Chemistry, 22.06.2019 09:40
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. an industrial chemist studying this reaction fills a 25.0l tank with 4.5 mol of sulfur dioxide gas and 4.5 mol of oxygen gas at 30.°c. he then raises the temperature, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 1.4 mol. calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 3

Chemistry, 22.06.2019 10:50
Determine the empirical formula for succinic acid that is composed of 40.60% carbon, 5.18% hydrogen, and 54.22% oxygen.
Answers: 1
You know the right answer?
Triphenylmethanol is insoluble in water, but when it is treated with concentrated H2SO4, a bright ye...
Questions

Mathematics, 08.09.2021 16:40

Mathematics, 08.09.2021 16:40

Computers and Technology, 08.09.2021 16:40


Mathematics, 08.09.2021 16:40

Mathematics, 08.09.2021 16:40

Biology, 08.09.2021 16:40



Mathematics, 08.09.2021 16:40

Mathematics, 08.09.2021 16:40

Mathematics, 08.09.2021 16:40

English, 08.09.2021 16:40

Physics, 08.09.2021 16:50