Chemistry, 21.04.2020 15:51 judyandaub1
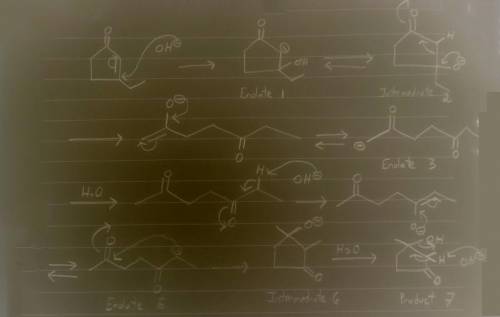
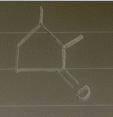
Aldol condensation of 2,5-heptanedione yields a mixture of two enone products in a 9:1 ratio. Treatment of the minor product with aqueous NaOH converts it into the major product; the interconversion proceeds as follows: Hydroxide ion adds to the double bond, forming enolate ion 1; Proton transfer occurs, yielding tetrahedral intermediate 2; Ring opening occurs, yielding enolate ion 3; Protonation of enolate ion 3 occurs, yielding 2,5-heptanedione; Deprotonation at C-6 occurs, yielding enolate ion 5; Enolate ion 5 attacks C-2, yielding tetrahedral intermediate 6; Protonation occurs to yield aldol addition product 7; Dehydration yields the more stable product.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 08:30
For each of the compounds below, show that the charges on the ions add up to zero. a. kbr b. cao c. li(2)o d. cacl(2) e. alcl(3)
Answers: 2

Chemistry, 22.06.2019 09:30
Which formula can be used to calculate the molar mass of hydrogen peroxide
Answers: 1


Chemistry, 23.06.2019 00:00
Predict the relative bond lengths of the three carbon-oxygen bonds in the carbonate ion (co2−3). what would you expect the charge to be on each oxygen? match the words in the left column to the appropriate blanks in the sentences on the right. make certain each sentence is complete before submitting your answer.
Answers: 3
You know the right answer?
Aldol condensation of 2,5-heptanedione yields a mixture of two enone products in a 9:1 ratio. Treatm...
Questions

Chemistry, 26.05.2021 20:30

Mathematics, 26.05.2021 20:30

Chemistry, 26.05.2021 20:30

Mathematics, 26.05.2021 20:30

Biology, 26.05.2021 20:30




Mathematics, 26.05.2021 20:30





Computers and Technology, 26.05.2021 20:30


Mathematics, 26.05.2021 20:30

Mathematics, 26.05.2021 20:30

Mathematics, 26.05.2021 20:30

Law, 26.05.2021 20:30

Mathematics, 26.05.2021 20:30