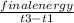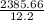Chemistry, 21.04.2020 08:47 sydneepollitt32
3. A student constructs a coffee cup calorimeter and places 50.0 mL of water into it. After a brief period of stabilization, the temperature of the water in calorimeter is determined to be 19.1 °C. To this is added 50.0 mL of water that was originally a temperature of 54.9 °C. A careful plot of the recorded temperature established T0 as 31.3 °C. What is the calorimeter constant (J/°C)?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 01:30
If 34.2 grams of lithium react with excess water, how many liters of hydrogen gas can be produced at 299 kelvin and 1.21 atmospheres? 2 li (s) + 2 h2o (l) yields 2 lioh (aq) + h2 (g)
Answers: 3

Chemistry, 22.06.2019 04:50
Compare the equilibrium constants for the systems shown in the table. which favors products the most? which favors products the least? rank these systems in order from most to least in terms of favoring products rather than reactants. d > b > a > c c > a > b > d b > c > d > a a > d > c > b
Answers: 1

Chemistry, 22.06.2019 08:40
Ageologist determines that a sample of a mineral can't be scratched by a steel nail but can be scratched by a masonry drill bit. based on this information, the sample mineral has to be softer than a. orthoclase. b. fluorite. c. apatite. d. corundum.
Answers: 2

Chemistry, 22.06.2019 21:30
Liquid ammonia is produced at high temperatures and under great pressure in a tank by passing a mixture of nitrogen gas and hydrogen gas over an iron catalyst. the reaction is represented by the following equation. n2(g) + 3h2(g) → 2nh3(g) changing all but one experimental condition will affect the amount of ammonia produced. that condition is a) increasing the concentration of both reactants b) changing the temperature within the tank c) decreasing the pressure within the tank. d) increasing only the amount of nitrogen present.
Answers: 1
You know the right answer?
3. A student constructs a coffee cup calorimeter and places 50.0 mL of water into it. After a brief...
Questions

Mathematics, 28.01.2021 04:00


Computers and Technology, 28.01.2021 04:00


Mathematics, 28.01.2021 04:00


English, 28.01.2021 04:00

Mathematics, 28.01.2021 04:00






Biology, 28.01.2021 04:00

History, 28.01.2021 04:00

Mathematics, 28.01.2021 04:00