
Chemistry, 21.04.2020 07:00 savannahworkman11
COLLEGE CHEMISTRY 35 POINTS
Determine the percent yield when 12 g of CO2 are formed experimentally from the reaction of 0.5 moles of C8H18 reacting with excess oxygen according to the following balanced equation:
2 C8H18(l) + 25 O2(g) 16 CO2(g) + 18 H2O(l)
Please show your work.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 18:50
Suppose you got a low yield of benzoin from your benzoin condensation reaction and thus only have 0.300 g of benzoin to use as the starting material for this reaction. how much concentrated nitric acid should you add? (concentrated nitric acid is 15.8 m). write your answer in the form x.xx ml
Answers: 1

Chemistry, 22.06.2019 05:00
If the density of water is 1.0 g/cm3, which of these materials would float in water, based on their densities? check all that apply. aluminum cork iron lead wax
Answers: 1

Chemistry, 22.06.2019 05:50
What happens when the temperature of a solution increases?
Answers: 2

Chemistry, 22.06.2019 10:30
What is the empirical formula of c6h18o3? ch3o c2h5o c2h6o c2h5o5
Answers: 1
You know the right answer?
COLLEGE CHEMISTRY 35 POINTS
Determine the percent yield when 12 g of CO2 are formed experiment...
Determine the percent yield when 12 g of CO2 are formed experiment...
Questions

English, 01.11.2019 20:31


Mathematics, 01.11.2019 20:31

Physics, 01.11.2019 20:31

Mathematics, 01.11.2019 20:31



Biology, 01.11.2019 20:31




Advanced Placement (AP), 01.11.2019 20:31


History, 01.11.2019 20:31


Mathematics, 01.11.2019 20:31

Biology, 01.11.2019 20:31


Chemistry, 01.11.2019 20:31

Mathematics, 01.11.2019 20:31

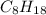 = 0.5
= 0.5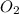 ⇒ 16 C
⇒ 16 C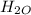
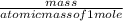
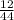
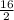 =
= 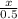
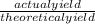 x 100
x 100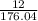 x 100
x 100

