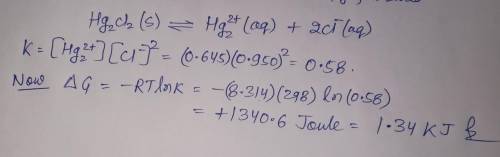Chemistry, 21.04.2020 04:51 micahwilkerson9495
A chemist fills a reaction vessel with 0.623g mercurous chloride(Hg2Cl2) solid, 0.645M mercury (I) (Hg2^2+)aqueous solution, and 0.905M chloride (Cl-) aqueous solution at a temperature of 25.0°C.
Under these conditions, calculate the reaction free energy ΔG for the following chemical reaction:
Hg2Cl2(s) ⇌ Hg2^2+ (aq) + 2Cl^- (aq)

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 14:00
What is the ph of a solution that has a hydrogen ion concentration of 1.0 * 10 -9 m?
Answers: 2

Chemistry, 22.06.2019 18:30
How many moles of bromine are needed to produce 3.23 moles of potassium bromide
Answers: 1

Chemistry, 22.06.2019 19:30
Estimate the molar mass of the gas that effuses at 1.6 times the effusion rate of carbon dioxide.
Answers: 1
You know the right answer?
A chemist fills a reaction vessel with 0.623g mercurous chloride(Hg2Cl2) solid, 0.645M mercury (I) (...
Questions


Mathematics, 28.10.2020 19:40

Mathematics, 28.10.2020 19:40



English, 28.10.2020 19:40

English, 28.10.2020 19:40

Mathematics, 28.10.2020 19:40



English, 28.10.2020 19:40


Biology, 28.10.2020 19:40

Business, 28.10.2020 19:40

Mathematics, 28.10.2020 19:40


Computers and Technology, 28.10.2020 19:40

Biology, 28.10.2020 19:40

English, 28.10.2020 19:40