Chemistry, 21.04.2020 00:24 vandonquisenberry
Measurements show that the enthalpy of a mixture of gaseous reactants increases by 215. kJ during a certain chemical reaction, which is carried out at a constant pressure. Furthermore, by carefully monitoring the volume change it is determined that -155. kJ of work is done on the mixture during the reaction. Calculate the change in energy of the gas mixture during the reaction. Round your answer to 2 significant digits. Is the reaction exothermic or endothermic?

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 09:20
What will most likely happen when two bromine atoms bond together?
Answers: 3

Chemistry, 22.06.2019 10:30
Which describes fat? a: a carbohydrate that produces energy b: a nucleic acid that directs cell function c: a lipid that stores energy d: a protein that speeds up a chemical reaction
Answers: 1

Chemistry, 22.06.2019 14:50
Given the following information: mass of proton = 1.00728 amu mass of neutron = 1.00866 amu mass of electron = 5.486 × 10^-4 amu speed of light = 2.9979 × 10^8 m/s calculate the nuclear binding energy (absolute value) of 3li^6. which has an atomic mass of 6.015126 amu. j/mol.
Answers: 2
You know the right answer?
Measurements show that the enthalpy of a mixture of gaseous reactants increases by 215. kJ during a...
Questions

Chemistry, 24.06.2019 17:10

English, 24.06.2019 17:10

Mathematics, 24.06.2019 17:10


Biology, 24.06.2019 17:10

English, 24.06.2019 17:10

Advanced Placement (AP), 24.06.2019 17:10





Spanish, 24.06.2019 17:10





Mathematics, 24.06.2019 17:10


History, 24.06.2019 17:10

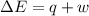
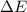 =Change in internal energy
=Change in internal energy