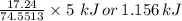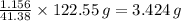Chemistry, 20.04.2020 22:22 albattatasraap5wymy
Dissolving potassium chlorate (KClO3) is even more endothermic than potassium chloride.
Your task is to determine how many grams of potassium chlorate you would have to add to 100 mL of water to produce the same temperature change as 5 grams of KCl.

Answers: 2
Another question on Chemistry


Chemistry, 23.06.2019 04:20
The graph shows one consequence of urban sprawl. how did urban sprawl contribute to the change in biodiversity
Answers: 2

Chemistry, 23.06.2019 04:40
6) (a) calculate the absorbance of the solution if its concentration is 0.0278 m and its molar extinction coefficient is 35.9 l/(mol cm). the depth of the cell is 5 mm. (b) what is the %t? (7) calculate the absorbance of the solution if the transmitted light intensity is 70% of the initial light beam intensity
Answers: 1

Chemistry, 23.06.2019 05:40
The independent variable in an experiment will be the variable that you o a) change ob) hold constant ng c) observe for changes
Answers: 2
You know the right answer?
Dissolving potassium chlorate (KClO3) is even more endothermic than potassium chloride.
Your...
Your...
Questions


English, 16.09.2021 14:00


Mathematics, 16.09.2021 14:00

Biology, 16.09.2021 14:00

Computers and Technology, 16.09.2021 14:00

English, 16.09.2021 14:00


Mathematics, 16.09.2021 14:00

Mathematics, 16.09.2021 14:00


Business, 16.09.2021 14:00


Social Studies, 16.09.2021 14:00


World Languages, 16.09.2021 14:00



Mathematics, 16.09.2021 14:00