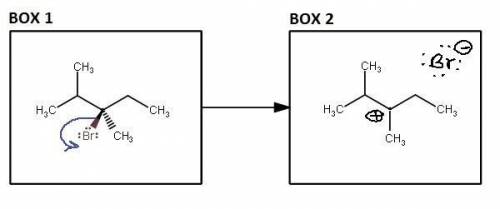
Draw the mechanism of the slow step that occurs in both first-order substitution and first-order elimination reactions for (R)-3-bromo-2,3-dimethylpentane in methanol with heat applied. Provide curved arrows in Box 1 to depict the flow of electrons and draw the intermediate in Box 2.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 23:00
What is the volume of the fluid in the graduated cylinder with accuracy and measured to the correct degree of precision? 41.2 ml 42.0 ml 41.23 ml 41.89 ml
Answers: 1

Chemistry, 22.06.2019 13:00
In what environment would mineral formation caused by high pressures and high temperatures most likely occur?
Answers: 3

Chemistry, 22.06.2019 19:30
Chlorine and water react to form hydrogen chloride and oxygen, like this: 2cl2 (g) + 2h2o (g) → 4hcl (g) + o2 (g) also, a chemist finds that at a certain temperature the equilibrium mixture of chlorine, water, hydrogen chloride, and oxygen has the following composition: compound concentration at equilibrium cl2 0.55m h2o 0.57m hcl 0.53m o2 0.34m calculate the value of the equilibrium constant kc for this reaction. round your answer to 2 significant digits.
Answers: 2

You know the right answer?
Draw the mechanism of the slow step that occurs in both first-order substitution and first-order eli...
Questions




Mathematics, 01.12.2020 22:40


Mathematics, 01.12.2020 22:40

Mathematics, 01.12.2020 22:40


English, 01.12.2020 22:40


Spanish, 01.12.2020 22:40



English, 01.12.2020 22:40

Mathematics, 01.12.2020 22:40

Mathematics, 01.12.2020 22:40

Health, 01.12.2020 22:40

Mathematics, 01.12.2020 22:40


English, 01.12.2020 22:40