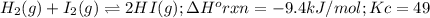H2(g), I2(g), and HI(g) are at equilibrium at a different temperature in a different vessel. (c) When the temperature in the vessel is decreased, does the equilibrium shift to the right, favoring the product, or to the left, favoring the reactants? Justify your answer. Rigid vessel

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 01:30
Arollercoaster car at the top of a hill has potential energy kinetic energy chemical energy light energy
Answers: 1

Chemistry, 22.06.2019 07:00
At 450 mm hg a gas has a volume of 760 l, what is its volume at standard pressure
Answers: 2

Chemistry, 23.06.2019 03:10
Which is true according to the law of conservation of energy
Answers: 1
You know the right answer?
H2(g), I2(g), and HI(g) are at equilibrium at a different temperature in a different vessel. (c) Whe...
Questions

Physics, 13.01.2021 16:20


Mathematics, 13.01.2021 16:20


Biology, 13.01.2021 16:20



Mathematics, 13.01.2021 16:20

Mathematics, 13.01.2021 16:20

Chemistry, 13.01.2021 16:20


Social Studies, 13.01.2021 16:20


Mathematics, 13.01.2021 16:20

English, 13.01.2021 16:20


Mathematics, 13.01.2021 16:20


Advanced Placement (AP), 13.01.2021 16:20