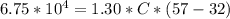Chemistry, 17.04.2020 21:34 diamondgdm
Calculate the specific heat capacity of a piece of ice if 1.30 kg of the wood absorbs 6.75×104 joules of heat, and its temperature changes from 32 ºC to 57 ºC.

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 09:00
Chen drew a diagram to compare the ways in which different organisms obtain nitrogen. which label belongs to the area marked z?
Answers: 3


Chemistry, 22.06.2019 18:00
Hydrogenation reactions, in which h2 and an "unsaturated" organic compound combine, are used in the food, fuel, and polymer industries. in the simplest case, ethene (c2h4) and h2 form ethane (c2h6). if 140 kj is given off per mole of c2h4 reacting, how much heat (in mj) is released when 12 kg of c2h6 forms?
Answers: 2
You know the right answer?
Calculate the specific heat capacity of a piece of ice if 1.30 kg of the wood absorbs 6.75×104 joule...
Questions



History, 10.11.2020 01:10

Arts, 10.11.2020 01:10


Mathematics, 10.11.2020 01:10

Mathematics, 10.11.2020 01:10







Mathematics, 10.11.2020 01:10

Mathematics, 10.11.2020 01:10

Chemistry, 10.11.2020 01:10


Mathematics, 10.11.2020 01:10

Physics, 10.11.2020 01:10

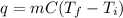 , where q is the energy in Joules, m is the mass in kg, C is the heat capacity, T_f is the final temperature, and T_i is the initial temperature.
, where q is the energy in Joules, m is the mass in kg, C is the heat capacity, T_f is the final temperature, and T_i is the initial temperature.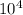 J, m = 1.30kg, T_f = 57, and T_i = 32. So:
J, m = 1.30kg, T_f = 57, and T_i = 32. So: