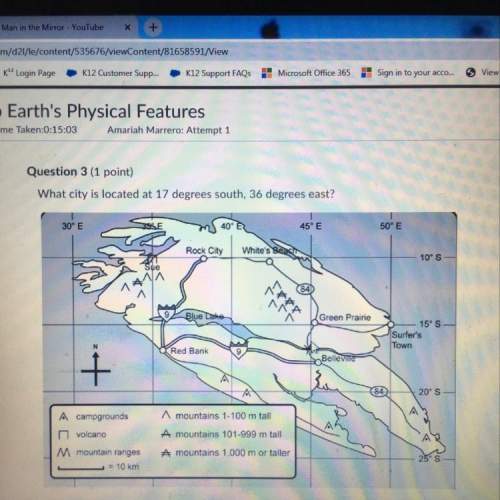
A solution is prepared by dissolving 0.23 mol of lactic acid and 0.27 mol of sodium lactate in water sufficient to yield 1.00 L of solution. The addition of 0.05 mol of HCl to this buffer solution causes the pH to drop slightly. The pH does not decrease drastically because the HCl reacts with the present in the buffer solution. The Ka of lactic acid is 1.36 × 10-3.
A lactate ion
B H2O
C lactic acid
D H3O+
E This is a buffer solution: the pH does not change upon addition of acid or base.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 03:30
Explain why pure hydrogen cyanide does not conduct electricity, but become a conductor when it is dissolved in water? (at room temp, pure hcn exists as a volatile liquid)
Answers: 1

Chemistry, 22.06.2019 14:00
8.98 dm3 of hydrogen gas is collected at 38.8 °c. find the volume the gas will occupy at -39.9 °c if the pressure remains constant.
Answers: 3

Chemistry, 23.06.2019 00:30
What is the percent by mass of magnesium sulfate in mgso4.7h2o
Answers: 3

Chemistry, 23.06.2019 03:30
If you need to add 27.50ml of a solution, which piece of glassware would you use to deliver this volume and explain how you would determine if the 27.50 ml was measured?
Answers: 1
You know the right answer?
A solution is prepared by dissolving 0.23 mol of lactic acid and 0.27 mol of sodium lactate in water...
Questions

English, 14.01.2021 01:20


Advanced Placement (AP), 14.01.2021 01:20

Chemistry, 14.01.2021 01:20

Mathematics, 14.01.2021 01:20

Mathematics, 14.01.2021 01:20

English, 14.01.2021 01:20



Mathematics, 14.01.2021 01:20

Arts, 14.01.2021 01:20


Mathematics, 14.01.2021 01:20




Geography, 14.01.2021 01:20



Mathematics, 14.01.2021 01:20