
Chemistry, 16.04.2020 00:50 jerenasmith77
What is the hydronium ion concentration of a 0.150 M hypochlorous acid solution with K a = 3.5 × 10-8? The equation for the dissociation of hypochlorous acid is:
HOCl(aq) + H2O(l) ⇌ H3O+(aq) + OCl-(aq).

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 05:50
What are transitions between a liquid and gas called? identify which way they are transitioning
Answers: 2


Chemistry, 22.06.2019 09:50
What are four significant sources of ghgs that come from wostem washington?
Answers: 2

Chemistry, 22.06.2019 14:30
How can carbon move from "land" to bodies of water? describe the way human impact has lead to increased levels of co2 in the atmosphere.
Answers: 2
You know the right answer?
What is the hydronium ion concentration of a 0.150 M hypochlorous acid solution with K a = 3.5 × 10-...
Questions


Mathematics, 02.12.2020 19:50


Mathematics, 02.12.2020 19:50

English, 02.12.2020 19:50

Mathematics, 02.12.2020 19:50

Mathematics, 02.12.2020 19:50


Physics, 02.12.2020 19:50





Biology, 02.12.2020 19:50




Mathematics, 02.12.2020 19:50

Mathematics, 02.12.2020 19:50

Mathematics, 02.12.2020 19:50

![[H_3O^+]](/tpl/images/0603/9545/6cfd2.png) is
is .
. be change of concentration.
be change of concentration.
![K_a=\frac{[A^-][H_3O^+]}{[HA]}](/tpl/images/0603/9545/2421d.png)
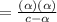
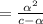
 [ For weak acid , the value of
[ For weak acid , the value of  ]
]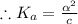
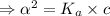
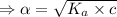


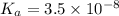
 M
M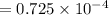 M
M

