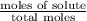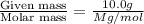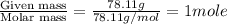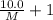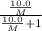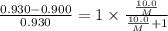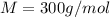Chemistry, 15.04.2020 19:55 gandalfhan
At a certain temperature, the vapor pressure of pure benzene (C6H6) is 0.930 atm. A solution was prepared by dissolving 10.0 g of a nondissociating, nonvolatile solute in 78.11g of benzene at that temperature. The vapor pressure of the solution was found to be 0.900 atm. Assuming the solution behaves ideally, determine the molar mass of the solute.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:40
How many electrons does silver have to give up in order to achieve a sido noble gas electron configuration?
Answers: 3

Chemistry, 21.06.2019 23:00
Layers of rock containing fossils, like the layers illustrated here, are most likely composed of rocks.
Answers: 2


Chemistry, 22.06.2019 14:00
How many absorptions would you expect to observe in the 13c nmr spectra of the following molecules? a) 3-chloropentane b) cis-4-methyl-2-pentene
Answers: 2
You know the right answer?
At a certain temperature, the vapor pressure of pure benzene (C6H6) is 0.930 atm. A solution was pre...
Questions


Mathematics, 24.02.2021 19:30


Mathematics, 24.02.2021 19:30


Mathematics, 24.02.2021 19:30

English, 24.02.2021 19:30

Biology, 24.02.2021 19:30


Chemistry, 24.02.2021 19:30

Mathematics, 24.02.2021 19:30





History, 24.02.2021 19:30



Biology, 24.02.2021 19:30

Geography, 24.02.2021 19:30

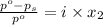
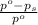 = relative lowering in vapor pressure
= relative lowering in vapor pressure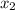 = mole fraction of solute =
= mole fraction of solute =