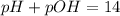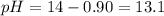Chemistry, 15.04.2020 17:23 tiffanybrown703
Suppose 50.0 ml of 0.350 m lithium hydroxide is mixed with 30.0 ml of 0.250 m perchloric acid. What is the ph of the resulting solution

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:40
Why did southern business leaders want to increase the number of slaves
Answers: 1

Chemistry, 22.06.2019 06:00
There are 6.022, 104 atoms of hg in 1 mole of hg the number of atoms in 45 moles of hg can be found by multiplying 4.5 by 6.022, 102 which is the number of atoms in 4.5 moles of hg, correctly written in scientific notation with the correct number of significant figures? 0 21,109 0 21,100 271, 1024 27.099, 100 mark this and retum save and exit submit
Answers: 1

Chemistry, 22.06.2019 13:10
What type of interaction occurs between the r groups of valine and isoleucine in a tertiary structure? view available hint(s) what type of interaction occurs between the r groups of valine and isoleucine in a tertiary structure? salt bridge disulfide bridge hydrogen bond hydrophobic interaction
Answers: 1

Chemistry, 22.06.2019 18:00
Hydrogenation reactions, in which h2 and an "unsaturated" organic compound combine, are used in the food, fuel, and polymer industries. in the simplest case, ethene (c2h4) and h2 form ethane (c2h6). if 140 kj is given off per mole of c2h4 reacting, how much heat (in mj) is released when 12 kg of c2h6 forms?
Answers: 2
You know the right answer?
Suppose 50.0 ml of 0.350 m lithium hydroxide is mixed with 30.0 ml of 0.250 m perchloric acid. What...
Questions

English, 21.04.2020 19:03





Mathematics, 21.04.2020 19:03


English, 21.04.2020 19:03

Biology, 21.04.2020 19:03

Biology, 21.04.2020 19:04




Mathematics, 21.04.2020 19:04


History, 21.04.2020 19:04



Biology, 21.04.2020 19:04

 .....(1)
.....(1)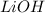 solution = 0.350 M
solution = 0.350 M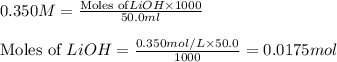
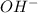
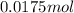 of
of 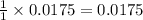 mol of
mol of 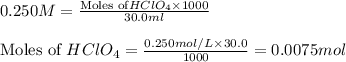
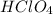 contains = 1 mol of
contains = 1 mol of 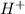
 of
of 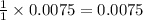 mol of
mol of 

![-log[OH^-]](/tpl/images/0601/9942/dd4c6.png)
![-log[0.125]=0.90](/tpl/images/0601/9942/574fd.png)