
If 4.168 kJ of heat is added to a calorimeter containing 75.40 g of water, the temperature of the water and the calorimeter increases from 24.58°C to 35.82°C. Calculate the heat capacity of the calorimeter (in J/°C). The specific heat of water is 4.184 J/g•°C Group of answer choices

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 23:00
Will mark brainliest26. which of these statements are true? (3 points)a. gases are compressibleb. gases fill their containers completelyc. the pressure of a gas is independent of the temperatured. gases have masse. gases exert pressuref. the pressure of a gas is dependent on the volumeg. gas pressure results from the collisions between gas particlesh. gases have a definite volume and shape
Answers: 1

Chemistry, 22.06.2019 22:30
Akno3 solution containing 51 g of kno3 per 100.0 g of water is cooled from 40 ∘c to 0 ∘c. what will happen during cooling?
Answers: 3

Chemistry, 22.06.2019 23:10
Afusion reaction takes place between carbon and another element. neutrons are released, and a different element is formed. the different element is a) lighter than helium.b)heavier than helium.c)the same weight as helium.d)dependent on the element that reacted with carbon.
Answers: 3

Chemistry, 23.06.2019 08:00
Amechanical wave that transports a lot of energy will have a
Answers: 2
You know the right answer?
If 4.168 kJ of heat is added to a calorimeter containing 75.40 g of water, the temperature of the wa...
Questions


Mathematics, 31.08.2019 10:50

Physics, 31.08.2019 10:50

Mathematics, 31.08.2019 10:50



Business, 31.08.2019 10:50

Chemistry, 31.08.2019 10:50

Social Studies, 31.08.2019 10:50

History, 31.08.2019 10:50

Chemistry, 31.08.2019 10:50







Mathematics, 31.08.2019 10:50

Mathematics, 31.08.2019 10:50

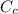 = 54.4
= 54.4 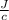
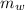 = 75.40 gm
= 75.40 gm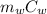 ΔT +
ΔT + 


