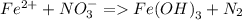Consider the following unbalanced redox reactions. In each case, separate the whole reactions into half-reactions, balance the half reactions, and combine to yield a balanced whole reaction. Compute the net reduction potential (E°) and pε° values for the net reactions and indicate whether the reaction is spontaneous as written. The reduction potential for Fe(OH)_3(s)/Fe^2+ is -0.181 V and pε is -3.08.
a. Fe^2 + + NO_3 = Fe(OH)_3(s) + N_2
b. Mn^2 + + Fe(OH)_3(s) = MnO_2(s) + Fe^2+

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:30
How much heat is released upon converting one mole of steam (18.0 g) from 100.0 ∘c to water at 25.0 ∘c? show work and constants, trying to figure out how it works. only given the heat capacity for steam and water so try to only use that
Answers: 1

Chemistry, 21.06.2019 23:00
Write a brief passage describing a neutral atom of nitrogen-14 (n-14). describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. use the periodic table to you. 14 protons and eletrons since its a neutral atom
Answers: 1


Chemistry, 22.06.2019 06:00
According to each substances heat of fusion, which of the items below requires more heat to be added per gram of substance to go from solid to liquid? silver sulfur water lead
Answers: 2
You know the right answer?
Consider the following unbalanced redox reactions. In each case, separate the whole reactions into h...
Questions







Mathematics, 11.02.2020 00:29


SAT, 11.02.2020 00:29

History, 11.02.2020 00:29

Biology, 11.02.2020 00:29





Biology, 11.02.2020 00:29