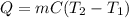In a coffee-cup calorimeter, 100.0 g of H2O and 100.0 mL of HCl are mixed. The HCl had an initial temperature of 44.6 oC and the water was originally at 24.6 oC. After the reaction, the temperature of both substances is 31.3 oC.
a. Was the reaction exothermic or endothermic?Explain.
b. Calculate how much heat the water lost or gained.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Often on a topographic map, every fifth contour line is darkened. what is this line called? a. key b.slope c.benchmark d. index contour
Answers: 1

Chemistry, 22.06.2019 01:40
Which characteristic of water it form droplets? a. low specific heat b. nonpolar structure c. high surface tension d. ability to dissolve substances
Answers: 1


You know the right answer?
In a coffee-cup calorimeter, 100.0 g of H2O and 100.0 mL of HCl are mixed. The HCl had an initial te...
Questions






English, 22.06.2019 23:00





Social Studies, 22.06.2019 23:00






English, 22.06.2019 23:00

Mathematics, 22.06.2019 23:00

Chemistry, 22.06.2019 23:00